INTRODUCTION Acute rhinosinusitis often accurs in both children and adults, and the main etiological agents are Streptococcus pneumonae, Haemophilus influenza and Moraxella catarrhalis (1). Though, other microorganisms can also be etiological agents in immune depressed patients (2).
Rhinosinusitis occurs from 25% to 30% of patients with AIDS (1). On those patients we can easily find infections by Pseudomonas aeruginosa, fungi, virus, parasites and even mycobacteria. Aspergillus is the main etiological agent of fungal rhinusinusitis, while Histoplasma capsulatum is rarely found, in both immune competent and immune depressed patients.
In this current study we reported a rare case of rhinosinusisits by Histoplasma capsulatum and discussed the aspects related to histoplasmosis patophysiology, clinical manifestations, diagnosis and treatment.
CASE REPORTA male patient, at his 40, was assisted at Ambulatório da Divisão de Clínica Otorrinolaringológica do Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (ENT Clinical Division Ambulatory of HCFMUSP) in January 2002 with nasal obstruction, yellow rhinorrhea, oedema and right eyelid hyperemia for three months. He also presented cacosmia, sudoresis in the afternoon, occasional fever and weighing 8 kg less during that period. He made use of several antibiotics with no improvement. He had quit smoking three years before, social drinker and reported pneumonia since six months. Although he denied cough, dyspnea, cutaneous alteration and odynophagia, he presented arthralgia on ankles and right wrist.
When examining, it was observed granulomatous-like mass on medium meatus of right nasal fossa and fistula draining yellowish secretion on right inferior eyelid. On CT from paranasal sinus and thorax could be respectively observe ethmoid hiding, maxillar, right nasal fossa (1,2), and diffuse interstitial infiltrate. It also presented HB=12.7 g/L (haemoglobin), HT=37.4 cel/mm3 (haematocrit) platelets = 157,000 and leukocytes =2600 cel/mm3, GOT (glutamic oxaloacetic transaminase) and GPT (glutamic pyruvic transaminase) respectively, 59 and 68 U/L. After serologies, it was found that patient was HIV and virus of hepatitis A and B carrier. CD4+ counting was 428cel/uL. The abdomen ultrasound showed hepatosplenomegaly.
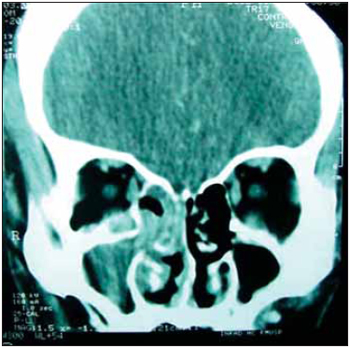
Picture 1. CT from paranasal sinus, coronal cut, presenting lesion on medium meatus and right maxillary sinus.
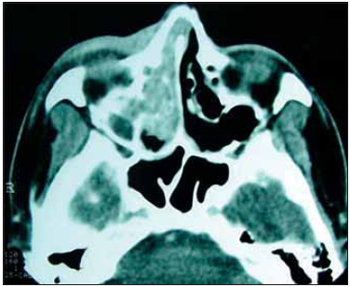
Picture 2. CT from paranasal sinus, coronal cut, axial cut, presenting lesion on medium meatus and right maxillary sinus.
It was done right maxillar sinus sinusoscopy with collection of a thick-whitish secretion and the cytological exam was negative. Partial result from maxillary sinus culture after 7 days was negative. Hemoculture, research of Resistant Alcohol-Acid Bacillus and rheumatological tests also had negative results. With necessity of a diagnosis to nasal mass, a biopsy from the lesion was done, and on histopathological exam, such lesion presented as a histoplasmosis on ethmoidal sinus and right medium meatus (3). The material culture obtained from sinusoscopy showed a growth of Histoplasma capsulatum 17 days after inoculation on Petry dish.
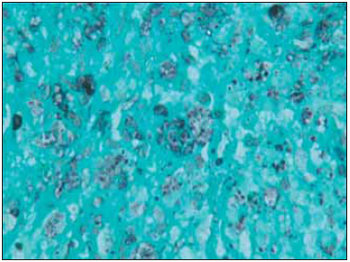
Picture 3. Picture 1000 times bigger, displaying histiocytes with plenty of round structures on cytoplasm, identified on impregnation by silver-staining (Gomori).
Patient was sent to Infectious Disease Service - HCFMUSP, There, antiretroviral therapy and Bactrim were introduced. Histoplasmosis was home-treated with itraconazole for six months. At the end of treatment, lesions on paranasal sinus could not be noticed anylonger. Patient developed with no nasosinusal signs or symptoms three year ago.
DISCUSSIONSamuel Taylor Darling was the first to describe Histoplasma capsulatum during his visit to Panama, 1904 (2). It concerns of a dimorphic fungus, found especially on the valleys of Mississipi and Ohio rivers (USA) and in South America. This fungus presents as mycelial shape in nature, and it is found in bird and bat excrements in the soil, especially in caves and hen-houses (2,4).
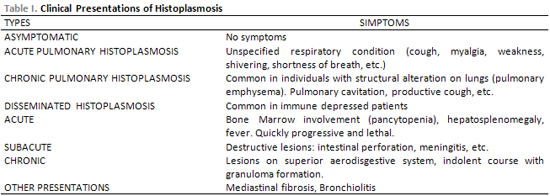
Mycelial spores are carried by the wind and when inhaled by man, they contaminate, but there is no human transmission (5). In body temperature, mycelial shape changes into leaven-like, so phagocyted by the macrophages on pulmonary alveoli. Macrophages expel the infection when able to destroy fungal cells through their enzymes, then patient presents no histoplamosis symptoms (6) (Table 1) Therefore, when cellular defense does not extinguish fungi, they increase in number inside macrophages (histiocytes this time) and are sent straight for endothelial-reticle system and for blood flow, causing infection spread through in all organs, especially on liver, spleen, bone marrow and adrenal gland (6).
As observed in other reports (1,7), our patient belongs to the HIV-positive group, and there are no case reports on sinusisits by histoplasm in normal-immune-system patients. HIV-positive patients present deficiency on cell immunity then sinusistis is more intense. It involves more sinuses and hardly responds to clinical treatment (1). The drop of CD4+ is related to frequency and seriousness of the infection (4). Atypical germes (Staphylococcus, Aspergillus, Rhizopus, Criptococus, Mycobacteria and cytomegalovirus), which are responsible for paranasal sinus infections, are the main organism found in these patients (3). About 80% of fungal sinusitis in HIV-positive patients is caused by Aspergillus (2). Stammberger et al (8) studying 48 cases of fungal rhinosinusitis observed the presence of Aspergillus in 39 patients. Therefore, Histoplasma capsulatum was responsible by alterations on paranasal sinus and on nasal fossa of our patient, what is rare, as only two cases are described in the literature (1,3). In the first case, paranasal sinus infection was caused by a process involving the hard palate (7) and on the second case, sinus contamination by Histoplasma capsulatum occurred through hematogenic pathway (1), as in our patient.
Histoplamosis can occur in different clinical ways (Table 1). The symptoms depend on immunological condition and fungal load inhaled. Most of people with normal immune systems develop either trivial clinical histoplasmosis or are totally asymptomatic. Unspecified breathing symptoms occur in an acute lung way (4) and the immune depressed patients (under cortisone therapy, lymphoproliferative neoplasies, AIDS) are more sensitive to develop them in a disseminated way, which can be deadly if not properly treated. The disseminated way can be acute, subacute or chronic. (5) The chronic diffuse way can be pointed out in our patient. The lesions on superior aerodigestive system are expressive in this case (8,9), where larynx and oral cavity lesions are frequent, opposing from the paranasal sinus, where, in our study, the lesion was. The more affected place is the tongue, followed by oral cavity and larynx (10). Goodwin et al (6), in their study, found 66% of patients with chronic disseminated way with oral lesions and 24% with laryngeal lesions.
Lesion on paranasal sinus is extremely rare and probably comes from hematogenic diffusion of Histoplasma capsulatum. From our case, we believe that organization changes and leukocytes drop, what is rare on chronic ways, are more probably caused by HIV infection than Histoplasm. Granuloma, with central necrosis, what was found in our patient, occurs in disseminated chronic way with more frequency. It is determined by painless lesions, and it should be distinguished from carcinoma and tuberculosis (10). Observed on thorax exam, the difference on pulmonary condition between pneumocystis and pulmonary histoplasmosis is expressively difficult to be distinguished (5). It much probably deals about the former by the absence of mass on mediastinum and lack of diffuse pulmonary condition on chronic disseminated way.
The colonization does not occur as it does on Candida or Aspergiluss. The Histoplasma capsulatum identification guarantees an exact diagnosis and the culture is the gold standard exam in order to confirm histoplasmosis, though it is limited by an incubation period from 2 to 4 weeks, what caused the tissue collection need for the histological study in our patient. The culture is not much used on severe cases, when intervention must be quick, besides culture on Sabouraud has been successful only in 25% to 40% (2). Culture is sensitive in disseminated and acute ways, but less sensitive in chronic ones, though aerodigestive lesion biopsies can be diagnozed if fixed in Gomori.
Histopathological exam from the material, which had its biopsy done and colored in Gomori (proper mean to observe Histoplasma), was used by a pathologist after a possible fungal process was suggested (3).This exam is less sensitive than culture and detection of antigens on blood and urine, what it can reach up to 75% of sensitivity (5).There are usually few organisms on granulomatous lesions and, in these cases, the use of Gomori is very important, as it was the invasive process done in order to obtain material to the study (biopsy).
Cutaneous test with histoplasmin was not done as it has epidemiological value and the number of false-negative on disseminated cases is high. Histoplasmin test presents negative results in 50% to 70% of the cases with disseminated way (4). Antigen detection by serological tests (complement fixation and latex agglutination) was not done in our case, however, it is a quick and sensitive method (90% of urine), and it is good to evaluate title drop of Histoplasma capsulatum, monitoring antifungal therapy (5). Complement fixation had been positive in 2/3 of the cases in disseminated way, and it is the best indirect diagnostic exam. In acute disseminated cases, where condition development is quickly progressive and lethal, this test is very useful.
There is no need of treatment for light cases. Symptomatic mechanical ventilation is more important in this stage. Anfotericin B is the treatment for disseminated cases, especially on those with AIDS, therefore, its nephrotoxicity and necessity of internation have limited its usage (11). In the last decade, the usage of itraconazol changed the condition of histoplasmosis treatment and, with its few side effects, oral use and antifungal efficiency, was distinguished on treatment of light and moderate disseminated cases and on chronic and acute pulmonary ways. On disseminated cases in quickly progressive HIV-positive patients, the use of anfotericin B for two weeks followed by a long-term period of itraconazol has been satisfactory (4). Its use in disseminated chronic cases from HIV-positive patients is very effective.
FINAL COMMENTSThe rhinosinusitis presentation showed the reason to diagnostic investigation and led to the finding of histoplasmosis on paranasal sinus and on infection by HIV. In this way, the ENT doctor must be aware of common agent diagnosis on rhinosinusitis etiology in immune depressed patients. Yet, s/he should be familiar with this entity, what should be included on differential diagnosis of possible causes of nasal obstruction.
Histoplasmosis requests high grade of suspicion, leading methods in the search of fungal infection, taking hold of invasive methods, such as endonasal biopsy. So that treatment is done in correct way. The pathologist should be aware of fungal origin of infection, guiding it on the histopathological exam.
REFERENCES1. Butt AA, Carreon J. Histoplasma capsulatum Sinusites. J Clin Microbiol, 35(10):2649-50, 1997.
2. Blitzer A, Lawson W. Fungal Infections of the Nose and Paranasal Sinuses. Otolaryngol Clin North Am, 26(6):1007-1013, 1993.
3. Schell WA. Histopathology of Fungal Rhinosinusitis. Otolaryngol Clin North Am, 33(2):251-276, 2000.
4. Bradsher RW. Histoplasmosis and Blastomycosis. Clinical Infectious Diseases, 22(Suppl 2):S102-11, 1996.
5. Kurowski R, Ostapchuk M. Overview of Histoplasmosis. Am Famy Physician, 66(12):2247-2252, 2002.
6. Goodwin RA, Shapiro JL, Thurman GH. Disseminated Histoplasmosis: Clinical and Pathological Correlations. Medicine, 59:1-33, 1980.
7. Lucatorto F, Eversole R. Deep mycoses and Palatal Perfuration with Granulomatous Pansinusitis in Acquired Immunodeficiency Syndrome: Case Reports. Quintessence Int, 24:743-48, 1993.
8. Stammberger H. Endoscopic surgery for mycotic and chronic recurring sinusitis. Ann Otol Rhinol Laryngol, 119(Suppl):S1-S11, 1993.
9. Sataloff RT, Wilborn A, Prestipino A, Hawkshaw M, Cohn J. Histoplasmosis of the Larynx. Am J Otolaryngol, 14(3):199-205, 1993.
10. Hiltbrand JB, Mcguirt WF. Oropharyngeal Histoplasmosis. Southern Med J, 83(2):227-31, 1990.
11. Kauffman C. Fungal Infections in Older Adults. Clinical Infectious Diseases, 33(15):550-55, 2001.
1. After graduating from the Division of Clinical Otorhinolaryngology Department of Ophthalmology and Otolaryngology da FMUSP.
2. Doctor-resident of the Division of Clinical Otorhinolaryngology Department of Ophthalmology and Otolaryngology da FMUSP.
3. Associate Professor of Discipline of Otorhinolaryngology, FMUSP.
Work done at the Division of Clinical Otorhinolaryngology, Hospital of the Faculty of Medicine of the University of Sao Paulo.
Mailing address: Bernardo Cunha Araujo Filho - Rua Oscar Freire, 1799 Flat: 1101 - Pines - Sao Paulo / SP - CEP 05409-011 - E-mail: bcaf@terra.com.br
Article received in on April 18, 2005. Article accepted with modifications on October 5, 2005.