INTRODUCTION AND OBJECTIVEHuman papilomavirus (HPV) belongs to the papillomavirus gender which belongs to the Papovaviridade family (1), made by circular, double-helix, non-enveloped DNA, with around 7200 to 8000 base pairs. It can lead to several benign, pre-malign and malign diseases in mucosas and skin surfaces. The HPV are evaluated according to their nucleic acids, that is, their genotype, in different kinds (2).
There are around 100 kinds of already-identified HPV (4) and, in terms of their malignity potential, they are classified into low (6, 11, 13, 32, 34, 40, 42, 44, 53, 54, 55 and 63 kinds) and into high risk of malignity (16, 18, 31, 33, and 35) (5). In some cases, specific kinds of HPV are related to certain clinical entities, for example: kinds 6 and 11 are the main ones involved in laryngeals papillomas and in most of condylomas of genital tract (6) while the 16, 18, 31 and 35 kinds are detected in most of cervix carcinomas(7).
The HPV persisting infection is considered the most important cause of cervix cancer (8), and its participation has also been implied in the development of other genital neoplasies such as vagina, vulva, penis, anus and oral cavity (9). The HPV is believed to be involved in 10 to 15% of human body neoplasies (4).
The sexual contact is the main way of HPV transmission (10), but there are other ways such as the maternal-fetus one (11), self-inoculation from skin lesions (12) or from genital lesions (13) and orogenital sexual abuse (14).
The HPV infections are not always macroscopically visible. Therefore, the HPV infection may be classified into: (a) latent, which can only be diagnosed through molecular biology methods; (b) subclinical, which does not have any clinical symptoms, but has subtle alterations which may be detected through diagnostical methods such as peniscopy, colpocitology, colposcopy and/or clinical biopsy (c), in which there are evident lesions at clinical analysis. From the molecular point of view, it is not known how an HPV infection remains latent and the other one, on the other hand, develops macroscopic lesion due to intense viral replication (4).
Most of HPV infections are asymptomatic - latent and subclinical, and the infection proportion is very small - from 1 to 2 % - which lead to apparent lesions. As well as other viral infections, the latent infections may be originated from: (a) virus exposure without leading to infection; (b) virus exposure with virus penetration in the host cell but without viral replication or without virus complete maturation (4).
In the anogenital tract, the HPV is the etiologic agent of genital warts or of also called acuminated condylomas, which are the most common benign genital tumors in both genders (15). Nowadays, there is no doubt that such lesions are transmitted through sexual contact, and it is recognizably the most frequent sexually transmitted disease (16).
Out of the 100 kinds of identified kinds of HPV, 24 of them have already been found in oral lesions (1, 2, 3, 4, 6, 7, 10, 11, 13, 16, 18, 31, 32, 33, 35, 45, 52, 55, 57, 58, 59, 69, 72 and 73 kinds) (17). In the oral cavity, the HPV is the etiologic agent of benign lesions such as scaly cell papilloma, focal epithelium hyperplasia, cancer and vulgaris wart (7).
The oral HPV predominance in macroscopically normal mucous membrane is very variable in the literature: it varies from 0 (18) to 81.1% (17), with na average of 10 % (19).
Bearing in mind the HPV importance as a sexually transmitted disease, its high predominance in the population (16) and its strong involvement as a cervix cancer agent (8, 19), it is essential to investigate other mucous regions, in addition to the anogenital region, which may lodge the HPV, such as the oral cavity.
The rising number of HPV anogenital infection and its high oncogenital potential make us think that other possible infection sites, little investigated as a routine or as a prevention, such as the oral cavity, may be recontamination nidus or even pre-malign and malign lesion development sites.
In the last decades, several authors (13, 20-28) focused their attention on HPV infection in other anatomic sites in parallel to anogenital infection, in order to understand if the anogenital infection caused by such virus may be predisposing factor for infection in other sites, such as the oral cavity.
Kellokoski et al (20), in 1990, performed prospective studies with 334 women with HPV genital infection in order to verify how frequently oral HPV would appear in those patients. The oral cavity has been examined at naked eye and abrasion of mouth vestibule mucous membrane has been performed bilaterally in 317 patients and, in 255 of them, biopsies for cytological and histological study have been performed. Oral lesions have been found in 127 patients out of 334 patients (38%), and the vulgar wart has been seen only in three patients (0.9%) and other lesions such as fibrous and papillary hyperplasia, candid infection, fissured tongue and leukoplakia have been found in the other patients. Koilocytosis has been found in 0.9% of the samples collected through abrasion and in 9.4% of the samples collected through biopsy.
Kellokoski et al (21), in 1992, performed a study in 262 women suffering from genital HPV in which they investigated HPV through abrasion, at naked eye, in mouth vestibule mucous membrane by using DB. HPV DNA has been found in 3.8% of the samples. There has been agreement of oral and genital HPV in 30% of the cases and the most frequent kind of HPV found in the genital and oral cavity has been the 6 kind.
Kellokoski et al (22), in the same year, performed oral biopsy at naked eye in 272 women suffering from genital HPV, and all of them were submitted to SB study and 85 of them to PCR. HPV DNA has been found in 15.4% and 29.4% of SB and PCR biopsies respectively. There has been agreement of oral and genital HPV in only 8% of the cases and the most frequent kind of HPV found in the genital and oral cavity has been the 6 and 11 kinds while in the genital region the 16 kind was found.
Van Doornum et al (23), in 1992, performed a study with 65 men and 111 women in whom an HPV research has been done in the oral and anogenital regions. In the oral cavity, the HPV investigation has been done through abrasion at naked eye and the material has been analysed through the PCR technique. HPV has been found in 24 men and in 32 women in the anogenital region, although oral HPV has not been found in any of them. There is no report of lesions frequency in the oral cavity.
Panici et al (24), in 1992, investigated HPV in the oral cavity of 66 women and 35 men with genital HPV. The oral cavity examination has been performed with a colposcope and acetic acid has been used as a marker of possible HPV oral lesions, followed by their biopsies. Through histological criteria, acuminate condyloma has been diagnosed in 49 out of 101 cases (48%) and HIS of oral cavity samples has also been done. Besides, HPV DNA has been found in 45% of the cases. In only eight patients there has been lesion which could be seen at naked eye, and in all of them acuminate condyloma has been confirmed through histological criteria. With the use of a colposcope, suspect lesions have been observed in 83 patients and, through histological criteria, condyloma has been confirmed in 46% of the cases.
Giraldo et al (25), in 1996, investigated HPV through cytological study in the oral mucous membrane of 51 women with genital HPV through abrasion, at naked eye. Conclusive cytological evidence of HPV infection has been considered due to koilocytosis, which has been present in 6% of the analysed oral cavity samples. There is no report of lesions frequency in the oral cavity.
Sarruf and Dias (26), in 1997, performed prospective study in which 54 patients, 13 women and 41 men with HPV genital infection, have been examined. All patients have been submitted to a clinical analysis at naked eye followed by mouth wash with acetic acid at 2% in order to identify white stains due to acetic acid. The material gathering for oral cavity cytological and histological study has been performed in the white stains due to acetic acid or, when they were not present, in the mouth vestibule mucous membrane bilaterally. Only patients with oral lesions have been submitted to their exeresis and the material has been submitted to the HIS technique in order to identify the HPV DNA. The oral cavity examination has been normal in 45 patients and White stains due to acetic acid have been found in nine patients. The cytological and histological evaluation have identified koilocytosis in seven patients, which corresponds to 13% of the cases. Oral lesions have only been found in two patients and the HIS study has identified HPV DNA in only one case.
Badaracco et al (27), in 1998, found oral HPV in 5 out of 10 women with genital HPV diagnosis through PCR and there was oral lesion in three of them. All patients have been submitted to oral cavity clinical analysis at naked eye and with colposcope, and the material has been gathered with abrasion of tongue dorsal and ventral faces or of previously identified lesion. There has been agreement of oral and genital HPV in 3 patients and the most frequent kind of HPV found in the genital and oral cavity has been the 16 kind.
Cañadas et al (28), in 2004, investigated HPV in the oral and anogenital regions of 166 women through abrasion and PCR. HPV oral and genital infection concomitance has been found in 12.7% of the cases, and the HPV 16 and 6 have been the most frequent ones in the anogenital and oral regions respectively. There is no report of lesions frequency in the oral cavity.
Smith et al (13), in 2004, investigated 165 pregnant women with genital HPV in order to investigate HPV in the oral cavity through PCR. The oral cavity has been examined at naked eye and the material has been gathered with saline solution mouth wash for 30 seconds. In 14 patients (8.5%) HPV has been found in the oral cavity, and the HPV 16 has been the most frequent one in the oral and genital regions. The kind of HPV has not agreed in any patient in both studied sites and there is not any report of lesion frequency in the oral cavity.
Taking into account the extreme result variability on articles found in the medical literature about oral HPV predominance on patients with anogenital HPV, a higher scientific investigation on this topic has become necessary.
The objective of this article is to determine how frequently HPV appears in the oral cavity of men with anogenital HPV confirmed through molecular biology.
MATERIAL AND METHODThe research project, including the free and clear Consent Term (Annex 1), has been approved by the Ethics and Research Committee on human beings from Fraternity of Santa Casa de Misericórdia de São Paulo (approval protocol number: 079/05).
The following inclusion criteria have been considered:
A) Male patients;
B) Aging from 15 to 60 years old;
C) Presence of one or more lesions in the anogenital area - penile corpus or glans, scrotum, inguinal, intrauretal or perianal/anal region - clinical or subclinical.
The following exclusion criteria have been considered:
A) Patients with fasting glucose higher than or equal to 126 mg/dL;
B) Patients with disease in the lymphoproliferative system such as leukemia and lymphoma, identified through the clinical history and/or complete hemogram;
C) Patients with HIV positive serologia;
D) Patients using immunossupressive drugs such as corticoisteroid, cyclosporin, azathioprine, tacrolimus, among others;
E) Patients whose molecular biology result of the anogenital lesion has been negative for HPV, despite of the compatible histological examination with acuminate condyloma.
In order to do the project, each patient has been previously informed, verbally, about the clinical and laboratorial exams to be performed. As a complement, the free and clear Consent Term was then given and signed, in case of agreement, by the patient and by the author.
Inicially, 10 patients from the Dermatology Ambulatory - Sexually Transmitted Diseases at Santa Casa de São Paulo have been selected.
The clinical and subclinical HPV lesions in the anogenital region have been submitted to excisional biopsy under local anesthesia with lindocaine at 2%. The warts have been considered the clinical lesions by the HPV in the anogenital region.
When macroscopical lesions did not exist, that is, anogenital clinical lesion, the colposcope has been used in order to perform the genitoscopy, which comprehended the peniscopy and the urethroscopy. The genitoscopy has been used in order to find white stains due to acetic acid in the anogenital region, corresponding to HPV subclinical legions.
The material gathered from the anogenital lesions has been divided into two fragments. One of them has been put into a dry Ependorf tube, which has been stored in freezer at -20ºC until it was sent to Ludwig Institute, for study through molecular biology. The other part of the material has been put in a tube containing formol at 10% and sent to the Pathology Department at Santa Casa de São Paulo, for histological study.
On the same day which the patients have been submitted to excisional biopsy of their anogenital lesions, their oral cavities have been examined at naked eye, with the help of artificial light from a photophore. Neither colposcope nor acetic acid application has been used during the oral cavity examination.
Once the oral lesion had been identified, its excisional biopsy has been done under local anesthesia with lindocain at 2%. The material gathered has been divided into two fragments. One of them has been put into a dry Ependorf tube, which has been stored in freezer at -20ºC until it was sent to Ludwig Institute, for study through molecular biology. The other part of the material has been put in a tube containing formol at 10% and sent to the Pathology Department at Santa Casa de São Paulo, for histological study.
When no lesion has been found in the oral cavity, the material gathering has been done through abrasion with steril brush (Libbs
® oncotic colpocitology gathering kit) of mouth vestibule mucous membrane bilaterally, dorsal and ventral tongue surface, hard and soft palate. The brush was then emerged into an Ependorf tube with PBS (buffered saline solution with phosphate) and the material was frozen in freezer at -20ºC until it was sent to the Ludwig Institute, for molecular biology study.
The material gathered through biopsy has been put into tubes with formol at 10%, processed with inclusion in paraffin, submitted to sections in rotating microtome, obtaining flaps measuring 4 micrometers thick. The flaps have been stained with hematoxylin and eosin and submitted to histological exam (Figure 1).
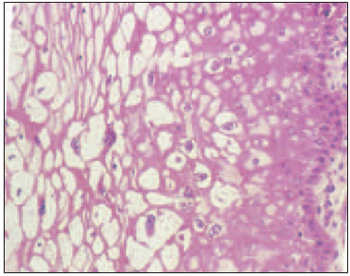
Figure 1. Condyloma histology - the HPV viral cytopatic effect, the koilocytosis, may be observed in this histological flap of condyloma lesion in the most superficial epithelium layers. They are cells which are surrounded by clear halo, picnotic nucleus and frequently of increased volume in the lesioned cell periphery.
Once the samples have been sent to the Ludwig institute, the material analysis through molecular biology has been done, which comprehended PCR and HR.
In order to amplify HPV DNA, the PCR with primer consensus MY09/11 has been used and for typing of different kinds of HPV HR "Line Blot" (LB) - Kit by Roche
® - Roche Linear Array has been used.
All patients have been applied to a questionnaire which can be seen on Annex 2.
Confidence Interval (CI) of 95% has been calculated for all estimations produced in this study.
Sensitiveness and specification tests have been performed in order to verify koilocytosis on HPV diagnosis, using the molecular biology exam as standard.
RESULTSAll patients' results may be seen on Tables 1 and 2.
All patients presented genital wart on physical exam, and in 7 of them the lesion was located in the penile corpus, in 2 of them in the anal region and in 1 of them in the penis glans.
The anogenital sample histological study made some acuminate condyloma suggestive findings evident in all of the 10 patients, which has been confirmed through molecular biology.
The HPV 6 has been the most frequently found kind in the anogenital region, appearing in 5 patients. The second most frequent kind was the HPV 11, found in 4 patients. The other kinds of HPV which have been found were: HPV 73, 16, 18, 42 and 52.
In terms of oral cavity, all patients presented normal macroscopical exam of oral cavity. The oral cavity abrasion for molecular biology study for HPV has been negative for all these patients.
The average age has been 31.4 years, varying from 21 to 50 years, with standard deviation 10.1.
In terms of race, 4 of them were dark-skinned, 5 of them were white and 1 was black.
In terms of addiction, 2 were smokers, 1 was alcoholic and none of them was drug addicted.
In terms of sexual partners, 8 reported being monogamist heterosexual, 1 polygamist heterosexual and 1 monogamist homosexual.
In terms of sexual activity frequency, the average has been 3.7 times a week, with standard deviation of 2.6.
When asked about their current partners, all of them denied pregressive or current oral lesions, 8 of them denied actual genital HPV and all of them denied pregressive HPV. When asked about their pregressive partners, 5 of them denied oral and/or genital lesion, 3 of them did not know about it and 2 of them reported genital lesions in previous partners, 1 of them known HPV.
Seven of them reported that they practice oral sex and 4 of them reported that they practice anal sex.
In terms of condom use, 4 of them answered "sometimes", 4 of them answered "never" and 2 of them answered "always".
IN terms of genital lesion personal background by any sexually transmitted disease, 3 of them stated having had genital HPV.
No patient, not even the ones with oral lesion which could be seen macroscopically, reported pregressive or current oral complaint.
DISCUSSIONThe kinds of HPV which have been found the mostly in the anogenital region of patients with clinical lesion have been the 6 kind (66.7%) and the 11 kind (23.3%) which are of low risk or benign (10). Those finds agree with the literature, which reports that those are the most frequent ones in anogenital condylomas (4). Syrjänen and Syrjänen (2000) (4) believe that the best HPV detection method is the PCR associated to other hybridization method in order to reduce the number of false-positive results, as it has been done in this study (PCR followed by reverse hybridization).
According to some authors (12, 13), the self-inoculation may be one HPV transmission way, that is, from skin or anogenital region infection, an infection in the oral cavity may occur. However, there is some discussion in the literature in terms of importance of such HPV infection propagation methods to the oral cavity (13).
In the last decades, several authors focused their attention on HPV infection in other anatomic sites in parallel to anogenital infection, in order to understand if the anogenital infection caused by such virus may be predisposing factor for infection in other sites, such as the oral cavity. From 1985 until 2004, few articles have been found in the literature about the anogenital HPV relation and its concomitance in the oral cavity (13, 20-28). Among those articles, there is no homogeneity in the technique used for HPV detection - cytology (20,24-26), dot blot (22) and PCR (13,21,23,27,28) and in the kind of oral and genital material gathering - abrasion (22,23,25-28), mouth wash (13) and biopsy (20, 21, 24), which makes the comparison of obtained results difficult. In the present study, we have used the molecular biology study with PCR and reverse hybridization and we have not found any patient with oral HPV. Studies which have also used PCR as HPV detection method, with (22) or without (13,23,27,28) other associated hybridization method, presented frequency of oral HPV varying from 0 to 50%. Consequently, the amount found in this study (0%) is not far from other values which have already been observed in the literature. However, as the latter ones show great variation, the comparative analysis with the results of this study becomes very complex.
Therefore, it is believed that the HPV anogenital infection presence is not a predisposition factor for oral HPV infection, which is suitable with the present study, in which there has not been any sample of positive HPV oral cavity.
From a practical point of view, such conclusion is of great importance for genital HPV patients, who are extremely worried about transmitting the virus to their partners and to other parts of their bodies.
As it is a pilot study, a higher clinical and scientific basis is necessary to establish oral and genital HPV relation and their implications in the urologist, gynecologist, proctologist e otorrinolaryngologist doctors daily clinics. It is necessary to raise the number of patients investigated in order to confirm the results obtained in this study.
CONCLUSIONThe oral HPV frequency in patients with anogenital HPV has been of 0% in this study.
BIBLIOGRAPHY1. Matthews REF. Classification and nomenclature of viruses. Intervirology 1982, 17:1-199.
2. Scully C, Prime SS, Maitland NJ. Papillomaviruses: their possible role in oral disease. Oral Surg 1985, 60:166.
3. Coggin JR, Zur Hausen H. Workshop on papillomavirus and cancer. Cancer Res 1979, 39:545-6.
4. Syrjänen KJ, Syrjänen SM. Papillomavirus infections in human pathology. England: John Wiley & Sons Ltd; 2000.
5. De Villers EM. Heterogeneity of the human papillomavirus group. J Virol 1989, 63:4898-903.
6. Reid R, Greenberg M, Jenson AB, Husain M, Willett J, Daoud Y et al. Sexually transmitted papillomavirus infections: the anatomic distribution and pathologic grade of neoplasic lesions associated with different viral types. Am J Obstet Gynecol 1987, 156:212-22.
7. Garlick JA, Taichman IB. Human papillomavirus infection of the oral mucosa. Am. J. Dermatopathol 1991, 13: 386-95.
8. Walboomers JM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 1999, 189:12-9.
9. Ringstrom E, Peters E, Hasegawa M, Posner M, Liu M, Kelsey KT. Human papilomavirus type 16 and squamous cell carcinoma of the head and neck. Clin Cancer Res 2002, 8:3187-92.
10. Zur Hausen, De Villiers EM. Human papillomaviruses. (Review). Ann Rev Microbiol 1994, 48:427-47.
11. Sedlacek TV, Lindheim S, Eder C, Hasty L, Woodland M, Ludomirsky A et al. Mechanism for human papillomavirus transmission at birth. Am J Obstet Gynecol 1989, 161:55-9.
12. Cohen BA, Honig P, Androphy E. Anogenital warts in children. Clinical and virologic evaluation for sexual abuse (see comments). Arch Dermatol 1990, 126:1575-80.
13. Smith EM, Ritchie JM, Yankowitz J, Wang D, Turek LP, Haugen TH. HPV prevalence and concordance in the cervix and oral cavity of pregnant women. Infect Dis Obstet Gynecol 2004, 12:45-56.
14. Yoshpe NS. Oral and laryngeal papilloma: A pediatric manifestation of sexually transmitted disease? Int J Pediatr Otorhinolaryngol 1995, 31:77-83.
15. Gissmann L, Zur Hausen H. Partial characterization of viral DNA from human genital warts (condiloma acuminata). Int J Cancer 1980, 25:605-9.
16. Carvalho JJM. Papilomavírus humano. In: Carvalho JJM. Manual prático do HPV: papilomavírus humano. São Paulo: Instituto Garnet; 2004. p.13-4.
17. Terai M, Takagi M, Matsukura T, Sata T. Oral wart associated with human papillomavirus type 2. J Oral Pathol Med 1999, 28(3):137-40.
18. Löning T, Meichsner M, Milde-Langosch K, Hinze H, Orlt I, Hörmann K et al. HPV DNA detection in tumours of the head and neck: a comparative light microscopy and DNA hybridization study. ORL 1987, 49:259-69.
19. Miller CS, Johnstone BM. Human papillomavirus as a risk factor for oral squamous cell carcinoma: a meta-analysis, 1982-1997. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001, 91:622-35.
20. Kellokoski JK, Syrjänen SM, Syrjänen KJ, Yliskoski M. Oral mucosal changes in women with genital HPV infection. J Oral Pathol Med 1990, 19:142-8.
21. Kellokoski JK, Syrjänen S, Chang F, Yliskoski M, Syrjänen K. Southern blot and PCR in detection of oral human papillomavirus (HPV) infections in women with genital HPV infections. J Oral Pathol Med 1992a, 21:459-64.
22. Kellokoski JK, Syrjänen SM, Yliskoski M, Syrjänen KJ. Dot blot hybridization in detection of human papillomavirus (HPV) infections in the oral cavity of women with genital HPV infections. Oral Microbiol Immunol 1992b, 7:19-23.
23. Van Doornum GJJ, Hooykaas C, Juffermans LHJ, Van Der Lans SMGA, Van Der Linden MMD, Coutinho RA et al. Prevalence of human papillomavirus infections among heterosexual men and women with multiple sexual partners. J Med Virol 1992, 37:13-21.
24. Panici PB, Scambia G, Perrone L, Battaglia F, Cattani P, Rabitti C et al. Oral condyloma lesions in patients with extensive genital human papillomavirus infection. Am J Obstet Gynecol 1992, 167(2):451-8.
25. Giraldo PC, Simões JA, Ribeiro Filho DA, Tambascia JK, Dias ALV, Pacello PCC. Avaliação citológica da orofaringe de mulheres portadoras do HPV genital. Rev Bras Ginec Obstet 1996, 18(3):737-42.
26. Sarruf MBJM, Dias EP. Avaliação citopatológica da cavidade bucal em pacientes portadores de infecção genital pelo papilomavírus humano (HPV). J Bras Doenças Sex Trans 1997, 9(2):4-18.
27. Badaracco G, Venuti A, Lonardo AD, Scambia G, Mozetti S, Panici PB et al. Concurrent HPV infection in oral and genital mucosa. J Oral Pathol Med 1998, 27(3):130-4.
28. Cañadas MP, Bosch FX, Junquera ML, Ejarque M, Font R, Ordoñez E et al. Concordance of prevalence of human papillomavirus DNA in Anogenital and oral infections in a high-risk population. J Clin Microbiol 2004, 42(3):1330-2.
1. Post graduanda in Otolaryngology (Mestranda the Department of Otolaryngology of Santa Casa de Sao Paulo).
2. Doctor of Medicine from the UNIFESP (Adjunct Professor of the Department of Otolaryngology, Faculty of Medical Sciences of Santa Casa de Sao Paulo).
3. Doctor of Medicine from the Santa Casa de Sao Paulo (Assistant Professor of Department of Surgery, Faculty of Medical Sciences of Santa Casa de Sao Paulo).
4. PhD in Medicine from the Santa Casa de Sao Paulo (Second Assistant Medical Clinic of Dermatology of the Santa Casa de Sao Paulo).
5. Masters in Medicine from the Santa Casa de Sao Paulo (PhD student in Otolaryngology at the Santa Casa de Sao Paulo).
Institution: Brotherhood of Santa Casa de Misericordia de Sao Paulo and Ludwig Institute for Research on Cancer-subsidiary Sao Paulo.
Sandra Doria Xavier
Mailing address: Street Nazareth Paulista, 163, ap 94B - Vila Madalena Sao Paulo / SP - CEP 05448000 - Phone: (11) 3666-6808 -- Fax: (11) 5082-2255 / 5082-2263 - E-mail: ssandoria@yahoo.com.br
This article was submitted in System Management Publications by R@IO em 20/10/2006 and approved on 21/2/2007 11:04:05