INTRODUCTION The synaptic model of neurocommunication in the Central Nervous System (CNS) ruled the neuroscientists for more than one century. With the appearance of the molecular biology and of the retaken of the concepts of neuronal plasticity in adults, it became clear the existence of alternative and economic mechanisms in the transmission of information. The non-synaptic diffusion neurotransmission (NDN), also referred to as transmission by volume, is being shown as the main mechanism in the transference of information in the CNS and presents an important role in the recovery of damaged areas(1,2).
The bilateral loss of the vestibular function or the bilateral vestibular reflex depression (BVRD), defined as the total lack of response of the vestibular system to the movement stimulus, results in multiple problems in the control of the posture and displacement, instability of pace and balance difficulties. Its most common etiology are toxicity by drugs, skull-encephalic injuries, meningitis, labyrinthitis, bilateral tumors, otosclerosis and several other factor, inclusively those associated with age(3). The main symptoms described by patients with BVRD are oscillopsia and unbalance. The first one is the "lack of sharpness" of the image derived from the loss of fixation of the object in the retina, resulting from the non operation of the vestibular and ocular reflex(4). But the unbalance increases in dark environments and irregular surfaces, as it is required at least two information systems to maintain the body balance. In the absence of a functional vestibular system, the CNS presents a difficulty to integrate properly the conflicting information between the visual and proprioceptive systems.
Up to current time, the preferred treatment for these patients is the vestibular rehabilitation (VR), which is efficient in up to 50% of the cases(5,6). Once the therapy is performed, the final result is definitive, there being, up to the moment, no other option to recover the possible residual limitations.
Despite the exclusion of the peripheral information, these patients keep the sensorial integration central mechanisms to maintain the postural stability. Thus, it could be possible to connect one artificial displacement receptor to the brain structures, associated with perception, integration and issuance of answers referring to the body balance, promoting, then, a reorganization of the cortical map and subsequent compensation of the damaged system(7). Once connected to the CNS through a man-machine interface (MMI), the artificial receptors may provide information and restore the functional regularity. Such receptors would be part of an equipment the information of which the brain accepts and controls as a natural part of its body(8,9). The MMI would provide to people with sensorial and motor damages a manner to use the CNS through artificial mechanisms in order to restore the lost abilities(10). This sensorial substitution process becomes possible thanks to the neuronal plasticity.
With this intention, Tyler, Danilov and Bach-y-Rita developed a vestibular substitution system and showed that the postural coordination can be restored using an MMI that employs an exclusive standard of electro-tactile stimulation on tongue's surface. This new biofeedback form was possible through equipment called BrainPort and it is based on the current concepts of NDN(11).
The BrainPort equipment transmits to the brain information about head's position (generally provided by the vestibular system) through a sensorial substitution channel: the tongue's surface. The use of the tongue as an optimal sensorial body is well established and grounded in its characteristics: nervous fibers' high density and sensitivity, in addition to the physical properties that provide reception and maintenance of the electric contact. In order the brain can properly interpret the information from a sensorial substitution device, it is not required that the information is presented the same way as the natural sensorial system. It is just required to code accurately the action potentials of an alternative information channel. With training, the brain learns to interpret properly the information and use it in accordance with the natural and ordinary perception data(12,13).
OBJECTIVE This study aims at assessing the efficiency of the BrainPort as a sensorial substitute of the vestibular organs in patients with BVRD who did not have a good response to the conventional vestibular rehabilitation.
CASUISTIC AND METHOD This is a clinic essay, previously judged and approved by the Ethics Commission for Research Project Analysis - CAPPesq of the Clinic Executive Board of Hospital das Clínicas of the Medicine School of Universidade de São Paulo (University of São Paulo/FMUSP).
The patients were selected from the Ambulatory of Otoneurology, of the Subject "Otorhinolaryngology" of the FMUSP after their explanation, acceptance and signature of the free and clarified agreement.
The subjects with body balance disturbance due to BVRD were included, and they should have concluded a treatment by RV without reaching a satisfactory result. The exclusion criteria were injuries in the oral cavity and tongue, tobaccoism, electric implantations such as heart pacing devices, neuro-degenerative diseases, and orthopedic injuries of inferior limbs.
The patients included in the study were assessed by their clinic history, Otorhinolaryngologic exam, exam of the skull pairs, balance and brain tests, electronystagmography, decreasing pendular rotatory test (DPRT) and by the protocol of the Sensorial Integration Test (SIT) of the Computerized Dynamic Posturography (CDP).
After the clinic characterization, the patients were, then, submitted to the intervention with the BrainPort equipment: Patients were offered tasks that present a progressive difficulty of posture change during the use of the equipment, which took 15 minutes per session, in two daily sessions held at 3 to 4 hours intervals. The training was delivered along 6 intercalated days (3 times per week, during 2 weeks) totalizing 12 sessions. The assessment of the clinic response to the treatment was made by the SIT of the CDP and by an analogous-visual scale (AVS) that characterizes the evolution of the symptoms pursuant to three criteria:
- remission (R): corresponding to 100% mitigation of symptoms.
- partial recovery (PR): corresponds to recovery of 50% to 90% of the symptoms.
- without recovery (WM): corresponds to a recovery percentage of symptoms below 50%.
For the analysis of the CDP (SIT) results, there has been considered the sensorial analysis and the balance index (BI).
Equipment There are two elements that integrate the BrainPort device:
(1) a 2x2 cm board attached to an accelerometer with 100 electrodes is placed on the surface of the tongue and detects the inclination and displacement of the head, in accordance with the body displacement;
(2) a device that has microcontrollers, sign processor, battery, timer and use control (Figure 1).

Figure 1. BrainPort Device: oral and controlling device.
The stimulation on the surface of the tongue is created by a sequence of pulses. The patient controls the tension level and one safety circuit monitors the outlet of stimulus, deactuating the system if the pre-defined current threshold is exceeded. The patient is able to turn on the equipment, adjust the stimulation intensity and centralize the stimulus in the electrode board using the controls.
The use of the equipment requires a instruction session in which the patient must learn how to use the stimulus perceived in the tongue and adjust his posture to keep it in the center of the board. The patient begins the training in the sit down or stand up position, with its head centralized and this position is recorded by a sensor that shall use it as the zero reference. At this moment, a stimulus is provided in the center of the board and it corresponds to the center of the tongue. With the displacement of the body, the stimulus also moves itself and the patient shall be instructed to keep the centralized stimulation - in the middle of the electrode board - as a response to its postural correction.
Training Protocol: DAY 1 - Assessment through CDP
- 1st session of 15-minute
- training 3-4 hours interval.
- 2nd session of 15-minute training
DAYS 2 to 6 - The procedure was repeated from the second to the sixth training day. At the end of the second session on the sixth day, the patients were submitted to an assessment by CDP and application of AVS.
The statistic analysis includes a drawing describing the cases.
RESULTS The sample comprised 5 cases of bilateral post-caloric vestibular reflex depression, 4 male patients and 1 female patient, with ages ranging from 54 to 74 years old (average of 60.6).
Besides not presenting a response to the bilateral post-caloric stimulation, all patients studied presented lack of response to the rotatory stimulus of the PRPD.
The etiologic diagnose and the age can be observed in Table 1.
The final balance index (BI) of the pre and post treatment CDP have its distribution represented in table 2, with an average of 34.8 in the pre-treatment stage and 61.0 in the post treatment stage.
The results of the clinic recovery with the proposed treatment using the BrainPort equipment are shown in Graph 1, in accordance with the AVS's criteria. The clinic recovery occurred in 100% of the cases, 2 with remission of the symptoms (40%) and 3 with partial recovery (60%) on the last day of the treatment.
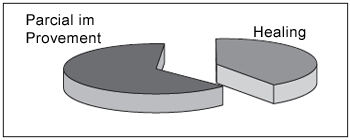
Graph 1. - Distribution of patients as to the clinical recovery after treatment with BrainPort.
The CDP data before and after the use of the BrainPort was also recorded and are reported here in Figure 2.
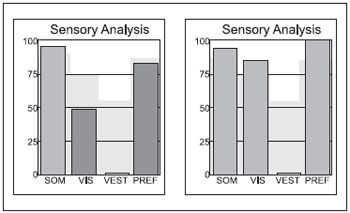
Figure 2. Sensorial analysis of patient 5 before and after the treatment with Brainport. SOM= somatosensory function; VIS= visual function ; VEST= vestibular function; PREF= visual preference. Note: The dark bars represent a performance below the ordinary standard for the age.
The BrainPort equipment appeared during the search for even more effective therapies in the treatment of vestibular diseases. The said equipment provides an electro-tactile stimulation on the surface of the tongue - sensorial substitution channel - and transmits to the CNS the information about head's positioning12,13. Probably, the most successful sensorial substitution system up to the present time is the Braille system, which allows "reading" information with finger tips, which are sensorial substitutes9,10. With the training, the subjects are conditioned to use the information coming from a sensorial substitution equipment, recovering information from a damaged system. Thus, the CNS is able to reorganize a sensorial damage or loss. All these said aspects are grounded on the current concepts of non-synaptic diffusion neurotransmission (NDN).
The bilateral vestibular reflex depression (BVRD) is a clinic condition that imposes several limitations and offers few recovery possibilities. Despite it is not frequent in patients with body unbalance, its morbidity required we pursue methods that, if not resolutory, at least offer to patients a better life quality. The limitations imposed on patients with BVRD include the lack of clarity of the image, which impairs the reading and driving activities. Further, they present an important unbalance in the pace, which takes away from them the firmness upon walking, impairing their displacement, especially in the external environments(5,6,14).
Our intention in this study was to assess this equipment's sensorial substitution capacity (BrainPort), improve the balance indexes in BVRD in patients already submitted to the conventional vestibular rehabilitation, as from which there is no professed treatment. We assessed in this study a sample of 2 cases of ototoxicity (40% of the sample), which is similar to the prevalence reported in literature(5,6). The others correspond to the traumatic, infectious and idiopathic etiologies, which make part of the etiologies responsible for the BVRD.
As to the assessment criteria, we used not only the electronystagmographic exam (ENG) but the PRPD. The most used exam in the investigation of the labyrinth dysfunctions is the ENG, which allows the analysis of the response of each labyrinth separately, but, however, it does not record the range of the physiologic frequency corresponding to the ocular-vestibular reflex. Thus, in order to record the low and high frequencies of the angular acceleration (0,1 to 1Hz), we used the PRPD that allows the assessment of the vestibular function in both sides(17). The PRPD is the gold standard for the diagnose of the bilateral vestibular losses, as it is possible to be absence of response to ENG, but with response present in other tests.
The CDP allows testing and quantifying the postural stability under several conditions, measuring the participation and interaction of several sensorial measurements (visual, vestibular and somatosensory). It assesses objectively the impact of the vestibular loss and selects the systems compromised in the maintenance of the balance. In BVRD, the largest loss is noticed under the 5 and 6 conditions, which are typically vestibular, with a quick loss of balance and falls. In the post treatment CDP example (Figure 2), there was no recovery of the vestibular system, but the potentialization of the visual and proprioceptive participation was critical for patient's clinic recovery, as well as the final increase of IE - 50(18).
In a previous study, when we assess the result of the Vestibular Rehabilitation (VR) in patients with BVRD, 87.5% of the subjects referred to a recovery and, even with different descriptions, they unanimously reported the increase of the body stability(19). In this new study, preliminary data suggests that the BrainPort acted efficiently as a sensorial substitute for the recovery of the body balance, exceeding the recovery previously obtained by the conventional RV both in the subjective assessment of the AVS, as in the objective assessment of the CDP.
The initial results evidence that the treatment is promising and we believe that new technologies can be developed in order to keep the sensorial stimulation, even in dynamic situations and upon body displacement. The study shall continue with the largest possible number of patients in order to get statistic data that can validate our hypothesis.
CONCLUSION Our preliminary data suggests that the BrainPort acted efficiently as a sensorial substitute in the recovery of the body balance, exceeding the recovery previously obtained by the conventional RV.
BIBLIOGRAPHY1. Agnati LF, Fuxe K. Volume transmission as a key feature of information handling in the central nervous system possible new interpretative value of the Turing´s B-type machine. Prog Brain Res 2000; 125, 3-19.
2. Sykova E. Extrasynaptic volume transmission and diffusion parameters of the extracellular space. Neuroscience 2004; 129, 861-876.
3. Brandt T. Vertigo, Its Multisensory Syndromes, 2nd Ed., Springer Verlag, London; 1999.
4. Leigh RJ, Zee DS. Tue Neurology of Eye Movements 3rd Ed., Oxford University Press, 1999.
5. Telian SA, Shepard NT, Smith-Wheelock M; Bilateral Vestibular Paresis: diagnosis and treatment. Otolaryngol Head Neck Surg 1991; 104:67-71.
6. Gillespie MB, Minor LB. Prognosis in bilateral vestibular hypofunction. Laryngoscope 1999; 109(1); 35-41.
7. Herdman SJ. Vestibular Rehabilitation. F.A. Davis Company, 2000.
8. Veraart C, Wanet-Defalque, MC. Representation of locomotor space by the blind. Percept Psychophys 1987; 42:132-39.
9. Arno P et al. Auditory coding of visual patterns for the blind. Perception, 1999; 28: 1013-1029.
10. Bach-y-Rita P, Kercel SW. Sensory substitution and the human-machine interface, Trends in cognitive Sciences, 2003; vol 7, 12: 541-46.
11. Tyler M, Danilov Y, Bach-y-Rita P. Closing and Openloop controlsystem; Vestibular Substitution through the thongue. J Integr Neuroscience, 2003. vol 2, 2:159-64.
12. Bach-y-Rita P. Tactile Sensory Substitution Studies. Ann NY Acad Sci, 2004; 1013:83-91.
13. Bach-y-Rita P, Kaczmarek KA, Tyler M, Garcia-Lara J. Form perception with a 49 point electrotatile stimulus array on the tongue: a technical note. J of Rehab Res & Develop., 1998; vol 35:427-30.
14. Bach-y-Rita P. Brain Mechanisms in sensory substitution. Academic Press, 1972.
15. Sargent EW, Goebel JA, Hanson JM, Beck DL. Idiopathic vestibular loss. Otolaryngol Head Neck Surg 1997; 116: 157-62.
16. Bach-y-Rita P. Nonsynaptic diffusion neurotransmission in the brain: functional considerations. Neurochemical Res, 2001. vol 26:871-73.
17. Kaplan DM, Marais J, Ogawa T, Kraus M. Does high frequency pseudo random rotational chair testing increase the diagnostic yeld of the electronystagmography caloric test in detecting bilateral vestibular loss in the dizzy patient? Laryngoscope 2001; 111:959-63.
18. Bles W, De Jong JMBV, De Wit G. Compensation for vestibular defects examined by the use of a tilting room. Acta Otolaryngol (Stockh) 1983;95;576.
19. Bittar RS, Pedalini ME, Ramalho JR, Carneiro CG. Bilateral vestibular loss after caloric irrigation: clinical application of vestibular rehabilitation. Rev Laryngol Otol Rhinol (Bord) 2005; 126(1):3-6.
1. Master on Medicine (Apprentice Doctor of the Otoneurology Clinic at HCFMUSP).
2. PhD on Medicine (Assistant Doctor of the Otoneurology Clinic at HCFMUSP).
Disciplina de Otorrinolaringologia do Hospital das Clínicas da FMUSP.
Correspondence: Camila de Giacomo Carneiro Barros/Depto de ORL do HCFMUSP (Otorrhinolaryngology Discipline at FMUSP Clinical Hospital) - Clinic of Prof. Ricardo Ferreira Bento
R. Dr. Enéas de Carvalho Aguiar, 255, 6º andar, sala 6021 - São Paulo/SP - CEP: 05403-000 - E-mail: camigcbarros@uol.com.br
This article was submitted to SGP (Publishing Management System) of R@io on May 27, 2007 and approved on August 12, 2007 at 20:07:55.