INTRODUCTION Acute otitis media (AOM) is an extremely common problem of public health among our children representing the most frequent type of infection in upper respiratory tract. It is associated to either high economical or social costs and it mainly affects preschool age children. By the end of the third year of life, 50% to 70% of children had at least one episode of OMA and by age 6, 75% of children had at least one or more episodes. Three or more episodes are reported between 9% and 18% of children during the first year of living and one or more episodes are reported in one third of all children during the first three years of life(1,2).
Otitis media is a very broad term. It encompasses a very large range of diseases from acute otitis media to chronic suppurative otitis media, passing through otitis media with effusion and recurrent otitis media. In Brazil, the Associação Brasileira de Otorrinolaringologia e Cirurgia Cérvico Facial (ABORLCCF), with its consensus about otitis, performed in 2002, has established as recurrent acute otitis media (RAOM) the disease of middle ear with three or more episodes in six months or four in twelve months with non symptomatic periods during the crisis(3).
Children with recurrent acute otitis media are classified with the expression otitis-prone condition. Many are the risk factors for disease recurrence like the first episode in the first year of life, family background of otitis, tube dysfunction, stay inday care centers, age, male gender, specific immune deficiencies(2,5,6). Although there might be an interaction between those factors and AOM, its relation is still a lot controversial, making some children prone to have recurrent acute otitis media(7).
Children with positive background of RAOM generally present alterations in the complement system and serum concentrations of certain immunoglobulin subclasses below the lower level like immunoglobulin G subclass 2 (IgG2) and subclasses 4 (IgG4), specific antibodies (IgG2) vs causing agents of acute otitis media(8,9,10,11). However other authors have not found the same results(12,13,14). Thus there is not a consensus in literature about the role of subclasses IgG on recurrent infections of upper respiratory tract specially on acute otitis media.
First reports on the relation between IgG subclasses deficiency and recurrent infections were done in 1968 and studies were about patients with recurrent infections who presented IgG subclasses below normality (15), with new reports in 1970 (16). Interest on this subject became wider with a study published in 1974 where there was a report of a family with serum levels of IgG2 and IgG4 below normality and recurrent and chronic infections of respiratory tract caused by Haemophilus influenzae(17).
In the 80's many researchers tried to reproduce studies described above but their results were very controversial. One of the possible proposed causes would be techniques used to measure immunoglobulines, previously measured with polyclonal antibodies and, with advent of new types of measurement, performed with monoclonal antibodies, more specific results were reached. Immaturity of immune system would be another cause for imprecise results since studies with longer segments showed that children with IgG2 dificiency turned to present normal levels of this immunoglobulin when adults. Besides absolute serum value of IgG2 below normality would not be responsible for recurrent otitis but selective deficiency of specific antibodies vs polysaccharide antigens of Streptococcus pneumoniae capsule would be the great responsible for recurrence of the disease(11,14).
This study is undertaken to describe immune role of immunoglobulin G subclass 2 and of specific antibodies in recurrent acute otitis media in childhood through systematic review of literature studies.
METHODOLOGY In order to select the articles an electronic search of literature on databases MEDLINE, LILACS and on The Cochrane Collaboration Controlled Trials Register was performed. The following search words were used: acute otitis media, immunology, recurrence, IgG deficiency, children. Finally bibliographic references of selected articles to be possibly included in this study were also reviewed in order to find articles not located with electronic search.
Studies that would evaluate deficiency of total immunoglobulinas and of subclasses IgG, specially IgG2, in patients with recurrent acute otitis media, with publishing date until December 2005 were included. In relation to the study drawings the following were included: studies of transverse and longitudinal cuts, with or without control groups, systematic reviews and metanalysis being useful only articles published in English, Portuguese, Spanish, Italian and French. To be included, the studies should present relation between acute otitis media and deficiency of IgG2 without other related factors as well as should contain samples with 20 or more patients. Evaluated individuals patients should be between 6 months and 18 years old. The samples should be clinical or populational based presenting patients with deficiency of IgG2 independent of gender, local of birth or laboratory analysis method.
Studies that related other etiologic factors with acute otitis media such as primary or acquired immune deficiency, allergic rhinitis, gastroesophageal reflux, craniofacial anomalies, hypertrophy of pharyngeal and palatine tonsils, tube dysfunction as well as case reports, letters to the editor, reviews from experts and articles with evaluated samples of adult patients or of deficiency of other immunoglobulines (IgA, IgG1, IgG3 ou IgG4). When duplicated articles were found only one of the versions was used.
The summary of the articles obtained through electronic search were examined by the author and by one more reviewer (MBB). First, a selection through the titles of the articles was performed excluding those that were about topics specifically not related with this review. Second, the summary of the articles were evaluated according to the criteria of inclusion and exclusion. In case there was no agreement among the reviewers about an article, a specific discussion about it was carried out until a final consensus. The reviewers were not blind for the names of the authors, institutions and publishing journals.
RESULTSTable 1 shows selection process of articles in several databases used. Only articles arising from 1982 fulfilled all the inclusion criteria. 127 articles were found in English, French, Spanish, Italian, Portuguese, German, Japanese and Chinese. However only articles in English, French, Spanish, Italian and Portuguese remained in the final selection. 09 of the 13 articles included in the review showed IgG2 measures below the normality in children with RAOM. 10 articles also evaluated selective deficiency of pneumococcal antipolysaccharide antibodies (IgG2) in children with recurrent infections of respiratory tract. 4 articles not included in the 13 original ones were associated to the immunization with antipneumococcal vaccine and its response on the recurrence of acute otitis media. Heterogeneity of methods found was great with varied technicians of measurement of total immunoglobulines and their subclasses, not showing a regular standardization among the articles finishing, thus, with distinct results.
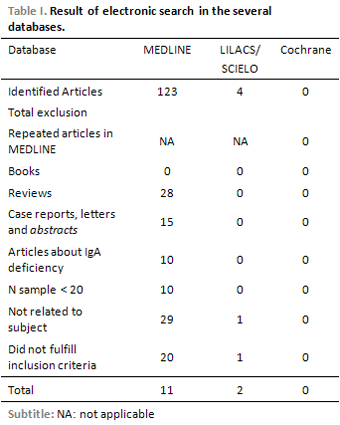
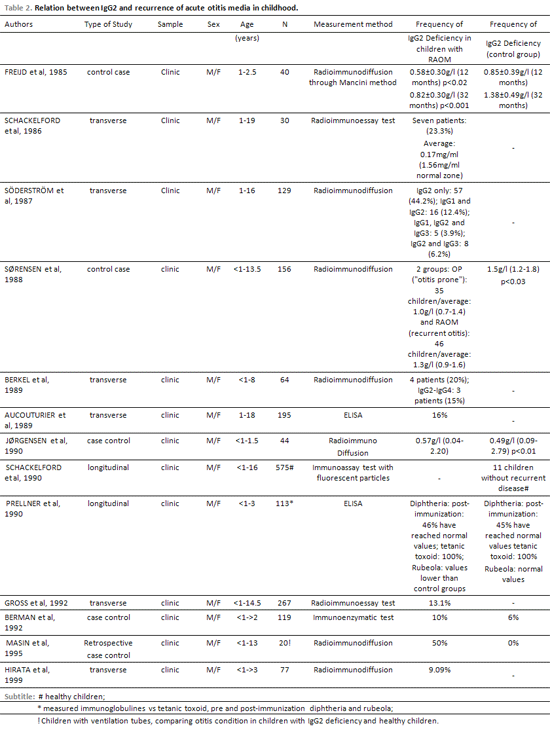
Table 2 shows the articles that have fulfilled inclusion criteria according to the relation between IgG2 deficiency and RAOM. 13 original articles, arising from 1985, associated recurrent otitis media with selective deficiency of IgG2(6,11,12,13,14,18,19,20,21,22,23,24,25). Five of the 13 evaluated studies showed control groups and eight were studies of transverse type. In relation to the studies with control groups only one has not shown correlation between IgG2 deficiency and the highest recurrence of AOM 14. Eight studies that have not shown control groups compared their studies with reference values of the method of each laboratory used in the studies. Five of them have found correlation between IgG2 deficiency and seriousness and severity of AOM. Nine of the 13 original studies have shown that children with RAOM are more likely to have IgG2 deficiency. Children below 12 months of life were included in every study. Among evaluated studies, incidence of IgG2 deficiency in children with RAOM varied between 9% and 44%.
Ten articles highlight the importance of specific antibodies determination vs certain bacterial agents such as pneumococcal and H. influenzae, showing that there children with RAOM are more likely to have selective deficiency of these antibodies(8,9,10,11,12,13,18,22,26,27).
DISCUSSION Acute otitis media is one of the most frequent childhood infections and one of the most common diagnosis in otolaryngology. In 1990, AOM was the secont most frequent diagnosis in all age groups in the United States with an average of 24 million visits(2, 28). It is the diagnosis that most induces the use of antibiotics in pediatric patients with estimated annual costs of 2.98 to 4 billion dollars(2,28,29).
Highest incidence of acute otitis media occurs in preschool children between 1 and 2 years of age. It is estimated that 50% to 75% of children will have experienced at least one episode of otitis media by the time they reach 3 years of age, and approximately 5% to 10% of them will have had recurrent infections of middle ear which deserves medical care and attention(2,26,30,31). During this period of life, it is also observed relatively low levels of immunoglobulines, specially subclasses IgG, that will increase in puberty.
Serum immunoglobulines are part of humoral response of immune system being the IgG, qualitatively, the most important of them. Serum levels of IgG in newborn child are identical or even higher than the mother's, showing active transport occurrence of immunoglobulines through the placenta. Such IgG levels acquired through placenta rapidly fall during the first 3 or 4 months of life as a result of maternal IgG catabolism determining very low IgG levels in children during the first 6 months of life. Due to this fall, a period of physiologica hypogammaglobulinemia is developed during the first 3 to 7 months of life. Besides this transitory reduction, IgG levels are the lowest ones among IgG subclasses during 6 months to 2 years of life.
Subsequently, IgG levels increase but each subclass shows a different period of augmentation. Minimum IgG level is approximately 25% of maternal initial level and it increases more slowly when compared to other subclasses. 50% of the levels in adults are not reached until 3 years of life and levels similar to adults' are only reached in puberty (after 15 years old). Nevertheless, immunoglobulin synthesis occurs immediately after the birth in response to gastrointestinal tract colonization, infections and other antigenic stimulation and becomes well-established after 6 months of life (15,16). Based on the evidences described about immunoglobulin production in childhood, children between 6 months and 2 years of life seem to be more susceptible to bacterial infections, thus, might also be more susceptible to recurrent infections of upper respiratory tract including acute otitis media.
In the last 3 decades, immunoglobulin quantitative determinations, including IgG subclasses, were performed in many serum evaluations of children and adults with positive background of recurrent infections of upper respiratory tract. In some studies, approximately 10% of evaluated serum showed deficiency of one or more IgG subclasses appearing to be an immunodeficiency not so rare as it was thought before(15,16).
The 13 original articles included in the final analysis of the study showed the same results found in review studies about IgG subclasses deficiency. Nine of them showed diminution in serum concentration of IgG2 in children with RAOM when compared to healthy children or to identical reference values of each laboratory. The length of the studies was similar with an average of 5 to 7 years also coincident with the literature data. However, in all studies, children with RAOM were between 6 months and 1 year old in the start of evaluation. These patients show a transitory IgG deficiency in this period what might significantly compromise the final result of the studies showing a false positive result. Incidence of IgG2 deficiency in children with RAOM varied between 9% and 44% among the sutdies. This so expressive variability may be explained by the floating levels of IgG subclasses according to age group, ethnic factor, infection background as well as evaluation methods. Different reference values of IgG subclasses were found in the analyzed studies. This fact is due to the variation of the used technician and geographic distribution of genetic markers that influenced their levels. Thus, each laboratory should determine its reference value according to the studied population.
In 10 studied articles of this study, reference to selective deficiency of IgG2 in relation to pneumococcal atigens was found. Children with RAOM background showed total IgG2 levels within normality with antipneumococcal IgG2 very below from the expected, though, showing the importance of selective evaluation of antibodies IgG2 in these patients. Therefore serum levels of total IgG2 within normality do not necessarily demonstrate an adequate immunological condition, making children with RAOM be likely to show a functional immunodeficiency. Thus, the use of a "functional antibodies" measurement is currently recommended, that is, measurement of antipneumococcal IgG and IgG2 antibodies of specific serotype in patients with recurrent infections background, specially RAOM. Measurement of the so-called "functional antibodies" has its own methodology and standardization and should only be performed in trustable laboratories that have a vast experience in these measures(32).
Most of the 13 evaluated studies date from 1990 and the most recent one, from 1999. A huge bibliographic review about the subject was carried out while the dissertation was being made and no more recent sutdy was found. A possible explanation would be a more specific interest in other immune deficiencies and even in antipneumococcal vaccine research as the subject is inserted in many of these articles. Most of literature is about immunological study of vaccines and genetic research not focusing on serum immunoglobulin determinations.
As a treatment for children with RAOM background the literature tells of antipneumococcal vaccine use that may appear with 7, 14 or even 23 types on antigens of most frequent serotypes of pneumococcus. Vaccines seem to be more efficient after 6 to 7 months of life because there is a transitory hypogammaglobulinemia, with the lowest levels of IgG2, during this period not showing satisfactory antibodies for immunization. After 10 years old, IgG2 levels are regularized and response to vaccine seems to be more efficcient. Some recent works do not observe any difference in the recurrence of acute otitis media after immunization with pneumococcal 23-valent vaccine opposing to most of authors 33,34,35. Therefore many studies are still necessary to reach a final conclusion about this vaccine on RAOM in childhood.
COMMENTS The 13 articles included in this study evaluated the relation of IgG2 with the recurrence of AOM in childhood. Nine (9) of them found a relation between IgG2 deficiency and RAOM. Incidence of IgG2 deficiency in children with RAOM varied between 9% and 44% among the researched sutdies, with very distinct variables.
Ten (10) studies showed the relation between IgG2 selective antibodies deficiency (antibodies vs polysaccharides of pneumococus capsule and of Haemophilus influenzae not typeable) and recurrence of AOM and of other infections of upper respiratory tract in childhood.
Clinical applicability of IgG subclasses measurement (absolute values), specially IgG2, as initial physical examination in children with RAOM would not be useful; measurement of "functional antobodies" is more recommended (IgGand IgG2 antibodies vs capsular polysaccharides of principal agents of AOM), specially in those patients where other risk factors were excluded ruled out.
BIBLIOGRAPHY1. Teele DW, Klein JO, Rosner B. Epidemiology of otitis media during the first seven years of life in children in greater Boston: a prospective, cohort study. J Infect Dis, 1989; 160(1):83-94.
2. Lanphear BP, Byrd RS, Auinger P, Hall CB. Increasing prevalence of recurrent otitis media among children in the United States. Pediatrics, 1997; 99(3):1-7.
3. Consenso sobre Otites Médias. Revista Brasileira de Otorrinolaringologia, 2002; 68(3 supl. 2):1-16.
4. Howie, VM, Ploussard, JH, Sloyer, J. The "otitis prone"condition. Am J Dis Child, 1975; 129(6):676-78.
5. Daly KA. Epidemiology of otitis media. Otolaryngol Clin North Am., 1991; 24(4):775-86.
6. Hirata CHW, Weckx LLM, Solé D, Figueiredo CR. Serum levels of immunoglobulins in children with recurrent otitis media. Invest Allergol Clin Immunol, 1999; 9(2):106-9.
7. Stenström C, Ingvarsson L. Otitis-prone children and controls: a study of possible predisposing factors. Acta Otolaryngol (Stockh), 1997; 117(1):87-93.
8. Umetsu DT, Ambrosino DM, Quinti I, Siber GR, Geha RS. Recurrent sinopulmonary infection and impaired antibody response to bacterial capsular polysaccharide antigen in children with selective IgG-subclass deficiency. N Engl J Med, 1985; 313(20):1247-51.
9. Rynnel-Dagöö B, Freijd A, Hammarström L, Oxelius VA, Persson MAA, Smith CIE. Pneumococcal antibodies of different immunoglobulin subclasses in normal and IgG subclass deficient individuals of various ages. Acta Otolayngol (Stockh), 1986; 101(1-2):146-51.
10. Herrod HG, Gross S, Insel R. Selective antibody deficiency to Haemophilus influenzae type b capsular polysaccharide vaccination in children with recurrent respiratory tract infection. J Clin Immunol, 1989; 9(5):429-34.
11. Shackelford PG, Granoff DM, Madassery JV, Scott MG, Nahm MH. Clinical and immunologic characteristics of healthy children with subnormal serum concentrations of IgG2. Pediatr Res, 1990; 27(1):16-21.
12. Jørgensen F, Andersson B, Hanson LA, Nylén O, Edén CS. Gamma-globulin treatment of recurrent acute otitis media in children. Pediatr Infect Dis J, 1990; 9(6):389-94.
13. Prellner K, Harsten G, Christenson B, Löfgren B, Heldrup J. Responses to rubella, tetanus and diphtheria vaccines in otitis-prone and non-otitis-prone children. Ann Otol Rhinol Laryngol, 1990; 99(8):628-32.
14. Berman S, Lee B, Nuss R, Roark R, Giclas PC. Immunoglobulin G, total and subclass, in children with or without recurrent otitis media. J Pediatr, 1992; 121(2):249-51.
15. Ochs HD, Wedgwood RJ. IgG subclass deficiencies. Annu Rev Med, 1987; 38:325-40.
16. Schur PH, Borel H, Gelfand EW, Alper CA, Rosen FS. Selective gamma-G globulin deficiencies in patients with recurrent pyogenic infections. N Engl J Med, 1970; 283(12):631-34.
17. Oxelius VA. Chronic infections in a family with hereditary deficiency of IgG2 and IgG4. Clin Exp Immunol, 1974; 17(1):19-27.
18. Freijd A, Oxelius VA, Rynnel-Dagöö B. A prospective study demonstrating an association between plasma IgG2 concentrations and susceptibility to otitis media in children. Scand J Infect Dis, 1985; 17(1):115-20.
19. Shackelford PG, Polmar SH, Mayus JL, Johnson WL, Corry JM, Nahm MH. Spectrum of IgG2 subclass deficiency in children with recurrent infections: prospective study. J Pediatr, 1986; 108(1):647-53.
20. Söderström T, Söderström R, Avanzini A, Brandtzaeg P, Karlsson G, Hanson LA. Immunoglobulin G subclass deficiencies. Int Archs Allergy appl Immun, 1987; 82(3-4):476-80.
21. Sorensen CH, Nielsen LK. Plasma IgG, IgG subclasses and acute-phase proteins in children with recurrent acute otitis media. APMIS, 1988; 96 (8): 676-80 22. Berkel AI, Barnett SL, Miller G, Jubelirer D, Bullard JW. IgG subclass deficiency in children with recurrent respiratory infections. J Okla State Med Assoc, 1989; 82(1):11-3.
23. Aucouturier P, Lacombe C, Bremard C, Lebranchu Y, Seligman M, Griscelli C, et al. Serum IgG subclass levels in patients with primary immunodeficiency syndromes or abnormal susceptibility to infections. Clin Immunol Immunopathol, 1989; 51(1):22-37.
24. Gross S, Blaiss MS, Herrod HG. Role of immunoglobulin subclasses and specific antibody determinations in the evaluation of recurrent infection in children. J Pediatr, 1992; 121(4):516-22.
25. Masin JS, Hostoffer RW, Arnold JE. Otitis media following tympanostomy tube placement in children with IgG2 deficiency. Laryngoscope, 1995; 105(11):1188-90.
26. Yamanaka N, Hotomi M, Shimada J, Togawa A. Immunological deficiency in otitis-prone" children. Ann N Y Acad Sci, 1997; 830:70-81.
27. Prellner K, Kalm O. Are there immunological or genetic markers that can predict recurrent acute otitis media? Ann N Y Acad Sci, 1997; 830:82-94.
28. Sih TM. Otite Média Aguda Recorrente. In: Tratado de Oto Rino Laringologia, São Paulo: Roca, 2002; p. 28-37.
29. Pappas DE, Hendley JO. Otitis media - a scholarly review of the evidence. Minerva Pediatrica, 2003; 55(5):407-14.
30. Pichichero ME, Casey JR. Acute otitis media disease management. Minerva Pediatrica, 2003; 55(5):415-38.
31. Cruz OLM, Souza MMA, Alvarenga EL. Estudo clínico de crianças com otite média aguda de repetição. RBM-ORL, 1998; 5(3):92-5.
32. Maguire GA, Kumararatne DS, Joyce HJ. Are there any clinical indications for measuring IgG subclasses? Ann Clin Biochem, 2002; 39(Pt 4):374-77.
33. Veenhoven R, Bogaert D, Uiterwaal C, Brouwer C, Kiezebrink H, Bruin J, et al. Effect of conjugate pneumococcal vaccine followed by polysaccharide pneumococcal vaccine on recurrent acute otitis media: a randomized study. Lancet, 2003; 361(9376):2189-95.
34. Mäkelä PH, Herva E, Sibakov M, Henrichsen J, Luotonen J, Leinonen M, et al. Pneumococcal vaccine and otitis media. Lancet, 1980; 2(8194):547-51.
35. Dhooge IJ, Van Kempen MJP, Sanders LAM, Rijkers GT. Deficient IgA and IgG2 anti-pneumococcal antibody levels and response to vaccination in otitis prone children. Int J Pediatr Otorhinolaryngol, 2002; 64(2):133-41.
1. Master degree on General Surgery, Otorrhinolaryngology sector, by Faculdade de Medicina da Universidade Federal do Rio de Janeiro (Rio de Janeiro Federal University Medical School) (medical doctor at Hospital Beneficência Portuguesa Petrópolis)
2. Head Professor of Otorrhinolaryngology of Universidade Federal do Rio de Janeiro (chief of the Otorrhinolaryngology clinic of Hospital Universitário Clementino Fraga Filho at Universidade Federal do Rio de Janeiro)
3. PhD Medical Doctor (Otorrhinolaryngology) by Universidade Federal de São Paulo (UNIFESP) (Otorrhinolaryngology Post-Graduation clinic chief at HUCFF/UFRJ)
4. Medical Residence on Plastic Surgery by Faculdade de Medicina da Universidade de São Paulo. Master Degree student by Hospital Heliópolis- SP (ENT surgery coordinator at Hospital São Lucas em Petrópolis)
Elisama Queiroz Baisch
Adress: R. Mal. Maurício José Cardoso, n. 105, apto 304 - Bloco 1 - Corrêas - Petrópolis/RJ - CEP: 25730-013 - Fax: (24) 22436114 - E-mail: elisbaisch@yahoo.com.br
This article was submitted to SGP (Publishing Management System) of R@io on April 5, 2007 and approved on June 29, 207 at 17:56:08.