INTRODUCTIONThe open cavity mastoidectomy is taken by many authors as the preferable surgical procedure for the treatment of cholesteatomatous otitis media, due to the high incidence of the diseases recurrence (1,2,3). This technique allows for a better removal of all the cholesteatoma and also a better post-operative control because it includes the aspiration of the disease's eventual recurrences (4). However, this unique cavity formation may cause some complications such as: deafness, persistent otorrhea, dizziness, poor aesthetics and the need for periodical cleaning of the cavity by the otorhinolaryngologist. Hence, in some selected cases we prescribe the elimination of the open cavity for the improvement of the patient's welfare. The techniques for open cavity elimination consist of reconstruction of the (MAE) external acoustic meatus rear wall, obliteration of the cavity or full ablation of the middle ear (5).
Since 1962, with the experiments of Mahoney et al. (6), which used the methylmetacrilate, several synthetic materials have been tried. The synthetic materials would offer the advantage of being easily obtained, in addition to the advantage of not submitting the patient to the risk of transmission of infectious diseases. In the other hand, they may present biocompatibility problems with risk of rejection.
A new material similar to hydroxyapatite has been widely studied for the reconstruction of osseous defections in orthopedic surgeries, neurosurgeries and bucomaxillofacial surgeries (7,8,9). It is the alpha-tricalcium-phosphate bone cement, that is an apatite carbonate formed by the mixture of the alpha-tricalcium-phosphate with calcium carbonate and monocalcium phosphate monohydrate powder. When these components are mixed to a solution of sodium phosphate, they form a crystalline structure similar to the bone mineral phase, the dahllite.
Although it has been used in surgeries for more than 10 years, there aren't studies about its application in mastoid cavity obliteration surgeries in the literature researched. The objective of this study is to evaluate the alpha-tricalcium-phosphate bone cement biocompatibility in animal model of mastoid cavity obliteration surgery.
METHODThis study was approved by the Ethics Committee for Analysis of Research Project (CAPPesq) of the Clinical Board of the Clinical Hospital of the Medicine College of the University of São Paulo. The experimental study was performed according to the manual about the laboratory animals cares. 20 young adult healthy animals of the species guinea pig (Cavia porcellus) of female sex, aged between 10 to 12 months and weight between 350 and 450 g were studied. Animals with signs of infection in the external or middle ear, pregnant and bearing congenital malformation were excluded.
The 20 animals were divided into 2 groups of 10 animals. Both groups were submitted to sterile surgical procedure and kept under observation for a period of 60 days. The animals were sacrificed after 60 days and the temporal bones were removed for histopathological study.
Surgical techniqueThe guinea pigs were anesthetized with a mixture of ketamine (40mg/kg) and xilasine (8mg/kg). Such anesthetic scheme allows for good anesthetic level for a period of 60 to 90 minutes without respiratory depression. All received the cefazolin antibiotic in the dosage of 50mg/kg during anesthetic induction.
After local infiltration with lidocaine at 2% with adrenalin 1:200.000, there was performed an incision up the auricular pavilion with exposure of the temporal muscle. Through the building of a muscle-periosteal piece lately pediculated, we exposed the temporal bone squamous part of the cortical. With the use of cutting and diamond drills and under constant irrigation, we removed the dorsal tympanic bulla cortical that allowed the full exposure of the internal side of this cavity (Picture 1). Afterwards the external acoustic meatus osseous rear wall is removed, which limits the dorsal tympanic bulla downwards (Picture 2). The cavity was then carefully cleaned and all the walls were scarified with the use of a diamond drill.
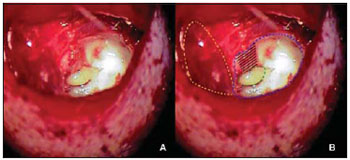
Picture 1. Left ear: (A) the tympanic bulla open. (B) scheme representation showing the tympanic bulla (dotted in blue), ossicular chain (dotted in green), periosteal muscle piece away (dotted in yellow) and posterior wall of MAE (striped in black).
Picture 2. Left ear: (A) tympanic bulla after removal of the MAE posterior wall; (B) scheme representation of the tympanic bulla open (dotted in blue) and the MAE osseous posterior wall removed (dotted in black) with exposure of the MAE skin.
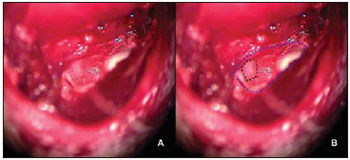
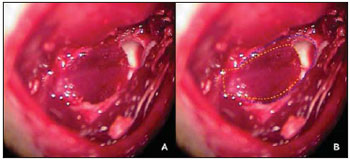
The muscle-periosteal piece was turned inside the tympanic bulla and covered the osseous defection of the external acoustic meatus rear wall (Picture 3). In the control group, 10 animals remained with the cavity without filling and in the study group 10 animals had the tympanic bulla obliterated with alpha-tricalcium-phosphate bone cement (Picture 4).
Picture 3. Left ear: (A) tympanic bulla open with periosteal muscle piece turning inside the tympanic bulla covering the osseous defection on the MAE posterior wall; (B) scheme representation of the tympanic bulla (dotted in blue) and muscular piece (dotted in yellow).
Picture 4. Left ear: (A) tympanic bulla obliterated with bone cement; (B) scheme representation of the bone cement obliterating the tympanic bulla (dotted in red) and base of the periosteal muscle piece (dotted in yellow).
The cement that was used comes conditioned in two recipients, a cup containing sterile calcium phosphate powder and a bottle containing sterile diluted sodium phosphate. The proportion between the powder and the solvent does not need to be calculated and it is enough to mix the total content of both bottles. The mixture must be applied and molded in up to 3 minutes, when the product hardening begins and is completed in 10 minutes.
The incision was sutured with the use of vicryl thread in the subcutaneous layer and nylon thread in the skin.
2.2. Histopathological material preparationAfter 60 days of the surgery, the animals were sacrificed. The animals were initially anesthetized with ketamine 160mg/kg and xilasine 16mg/kg, and then received lethal injection of KCI 19.1% intracardiac.
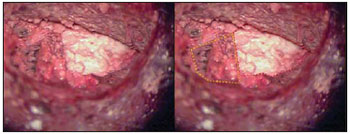
The guinea pig temporal bone was removed with the use of scope, hammer and scissors. The piece was removed carefully to maintain the tympanic bulla and the cement intact (Picture 5).
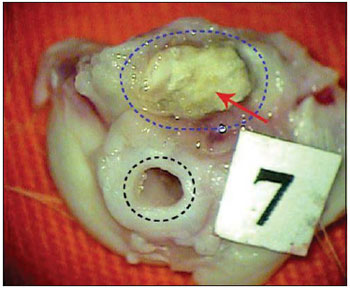
Picture 5. Guinea pig's left temporal bone removed. Bone cement (red arrow) obliterating tympanic bulla (dotted in blue) and MAE (dotted in black).
The material was then fixed with alcohol at 70% and included in methylmetacrilate blocks. The blocks were cut with low speed diamond slitting wheel (Isomet-Buehler) with thickness of 1 mm. Then the cuts were polished to achieve the thickness of 10 µm, with the use of a metallographic polisher. The cuts were finally colored with 0.1% of toluidine blue and forwarded to the pathologist.
2.3. Data analysis The histopathological analysis was carried out by a pathologist of the Pathology Department of the Clinical Hospital of São Paulo, who used an optical microscope. The blades were initially examined in a panoramic increase for the identification of all fields and then in increases of 12.5, 40, 50, 200 and 400x.
The clinical analysis was made before the temporal bone removal. The skin and muscular piece evaluation was performed with macroscopic exam and the otoscopy with the use of surgical microscope.
The guinea pigs were studied as for the presence of material rejection signs, grade of ossification and occurrence of complications.
Biocompatibility analysisFor the material biocompatibility analysis there were evaluated clinical and histological data of the material.
In the clinical evaluation the following data were collected:
Presence or absence of granulomatous inflammatory reaction signs in the surgical site. Presence or absence of material extrusion signs.As a histopathological parameter for the material biocompatibility evaluation, we studied the presence or absence of granulomatous inflammatory response with giant cells. For the analysis of inflammatory reaction and formation of foreign body reaction a classification in 5 grades was created (Board 1).
According with the grade of inflammatory response determined by the pathologist in the histological analysis, we divided the patients into 3 types (Board 2).
Ossification grade analysis For the mastoid cavity ossification grade analysis we created a classification in 4 grades (Board 3).
By using such classification the pathologist classified the ossification grade in the control and cement groups.
Analysis of complications incidence to the procedure
For the complications incidence analysis, the guinea pigs were evaluated as for the presence or absence of the following parameters:
Death of the animal. Effusion in the middle ear. Signs of skin or muscle infection. Signs of skin or muscle necrosis. Material extrusion. MAE stenosis. Cutaneous Fistula.Statistical analysisThe calculation of the sample size was made by taking as 0% the incidence of foreign body reaction or cavity chronic inflammation in guinea pigs submitted to mastoidectomy without its obliteration, at a level of significance of 5% and power (error type II) of 80% in a one-tail hypothesis test. 10 guinea pigs were necessary in each group (study and control) to detect a minimal difference of 6.5% of incidence of foreign body reaction or cavity chronic inflammation after the obliteration with alpha-tricalcium-phosphate bone cement.
The qualitative variables were described by means of their frequency and the semi-quantitative variables of their averages and standard deviations. For comparison of the different grades prevalence of inflammation, ossification and complications incidence between the groups, the chi-square test and the Fisher's exact test were used. The averages of scores ascribed to each ossification grade were also compared between the groups by using the Mann-Whitney U non-parametrical test. A level of significance of p<0.05 was admitted.
RESULTSThe control group animals initial weight average was of 396.8 g and the final weight average was of 570 g. The control group weight gain average was of 173.3 g during the study period. The weight gain percentage on the initial weight in the period was of 43.7 %.
The cement group animals initial weight average was of 398.8 g and the final weight average was of 574.3 g. The cement group weight gain average was of 175.5 g during the study period. The weight gain percentage on the initial weight in the period was of 44.0%.
In both groups, the death incidence after the procedure was of 2 (20.0%) animals in the first post-operative day (p = 1.0), probably for anesthetic complications because they occurred in the first post-operative period and were equal in both groups. The remaining animals clinical analysis did not identify any sign of other complications - skin and muscle infection, middle ear effusion signs, external acoustic meatus stenosis, cutaneous fistula or formation of secondary cholesteatoma - in any sample of the control and cement groups. The guinea pigs clinical analysis did not observe signs of granulomatous inflammatory reaction around the cement in any cement group animal. There wasn't either any case of material extrusion.
After the temporal bones removal, we observed that in all the cement group samples the material was firmly adhered to the mastoid cavity.
The histological analysis of inflammatory reaction grade and type in the cement and control groups are described in Table 1. There was no sign of foreign body type inflammatory reaction in the cement group with 8 (100%) of the samples presenting grades 1 and 2 inflammatory reaction. Only 3 animals of the control group (37.5%) had type 3 inflammatory reaction, while all the other guinea pigs (100% of the cement group and 62.5% of the control group) had type 1 inflammatory response. In spite of the difference presented in the inflammatory reaction types prevalence in the groups, such difference was not statistically significant (p=0.2).
As for the ossification grade study, the control group presented ossification of grade 3 and 4 (50%) of the samples and grade 4 (50%) in 4 samples studied. In the cement group we observed an ossification of grade 1 in 1 (12.5%) sample and grade 2 in 7 (87.5%) samples (Table 2).
By ascribing the weights to each animal according to the ossification grade, we observe that the control group average ossification grade (3.5) was larger than that verified in the cement group (1.875) (Chart 1). The control group presented in a statistically significant manner (p < 0.001) more intense grades of ossification (average 3.5 ± 0.5) than the cement group (1.9 ± 0.4).
DISCUSSIONThe guinea pig (
cavia porcellus) has a temporal bone cellular aeration composed by ventral and dorsal tympanic bullas and the retro-tympanic cells. As shown by an analogy with the human temporal bone, the dorsal tympanic bulla would be equivalent to the epitympanic and attic recess. It is located in the upper rear portion of the tympanic cavity and relates to the upper intracranial cavity, with the petromastoid part of the temporal bone in the middle and with the tympanic cavity and the external acoustic meatus in the lower part. The communication between the dorsal tympanic bulla and the tympanic cavity is made through a hiatus located in its lower portion, in which the incudo-malleolar complex is projected (10,11). Upon removal of the external acoustic meatus upper wall, the tympanic bulla communicates with the external acoustic meatus and forms a single cavity, similar to that found in the open mastoid cavities.
Several types of materials have been used for the open mastoid cavity obliteration. The mostly employed materials are the autolog ones, such as cartilage, bone, fat, muscle and bone mass that offer the advantage of not undergoing rejection and may be obtained during the surgery (5,12,13). The disadvantage is the difficulty to obtain material in a sufficient quantity for the procedure.
Then, since 1962, with the experiments of Mahoney et al. (6), which used the methylmetacrilate, several synthetic materials have been tried. The synthetic materials would offer the advantage of being easily obtained, in addition to the advantage of not submitting the patient to the risk of transmission of infectious diseases. In the other hand, they present problems as for the biocompatibility with high levels of rejection.
Among the synthetic materials that were studied for this purpose, we may mention silicon, proplast, ionomeric cement and bioactive glass cement (14,15,16,17).
The silicon, proplast and ionomeric cement are not used anymore due to the incidence of complications and the bioactive glass cement is still in experimental study phase (18).
More recently, calcium and phosphate based synthetic materials have been used and they present a biochemical structure similar to the human bone mineral phase. The calcium and phosphate aloplastic implants are those composed by calcium and phosphate in apatite shape. The apatite is composed in crystal shape that has a chemical structure represented by the formula Ca5(X)(PO4)3. In this structure the position X may be occupied by ions fluoride (F), chloride (CI), hydroxide (OH), or half carbonate (CO3) (19).
The first material of this studied group was hydroxyapatite. Such material has been offering good results when used in the MAE reconstruction or mastoid cavity obliteration (20,21,22). As of the study of hydroxyapatite, new types of materials were developed.
The alpha-tricalcium-phosphate bone cement is a product composed by 3 solutions: alpha-tricalcium-phosphate, calcium carbonate and monocalcium phosphate (23). When these components are mixed to a solution of sodium phosphate, they form a crystalline structure of apatite carbonate similar to the bone mineral phase, the dahllite (24,25).
The material time of preparation, which consists of a solid phase mixture with the solution of sodium phosphate, is of approximately 2 minutes, and the time for the material manipulation is of 5 minutes. After placement of the material there occurs its hardening with the formation of dahllite. The time of hardening in the normal corporeal temperature is of 10 minutes (24,26,27). After 12 hours, between 85 to 90% of the material is dahllite and its maximal compressive force is of 55 megapascals (24). Constantz et al. studied the dahllite biochemical composition and its crystalline structure formed in this reaction and confirmed that it's similar to the dahllite naturally present in the bone (24). This material has been widely used in the filling of osseous defections in orthopedic surgeries, neurosurgeries and bucomaxillofacial surgeries. Studies in ossicular chain reconstruction surgeries in guinea pigs with the alpha-tricalcium-phosphate cement showed good results with low levels of inflammation and foreign body reaction (28,29).
For the biocompatibility study the samples were studied by using clinical and histopathological parameters. The classification in 5 grades of inflammatory reaction used in this study for the measuring of inflammatory reaction grade was created from the adaptation of the classification used by Hoffman et al., who studied the inflammatory reaction grade in ossicular chain reconstruction surgery in guinea pigs with the use of alpha-tricalcium-phosphate cement (28). Such classification as used by Hoffmann et al. was divided into 4 grades. Because this classification was inadequate in the cases of presence of acute inflammation reaction, we added a fifth grade (grade 5). From this classification in 5 grades, we formed a subdivision in 3 groups we called types 1, 2 and 3. The type 1 corresponds, histopathologically, to the presence of inflammatory reaction presence that normally occurs in tissues submitted to surgical trauma and involves grades I and II (Picture 6). Type 2 corresponds, histopathologically, to the presence of foreign body type inflammatory reaction and comprises grades III and IV. Type 3 corresponds, histopathologically, to the presence of acute inflammatory reaction with possible infection.
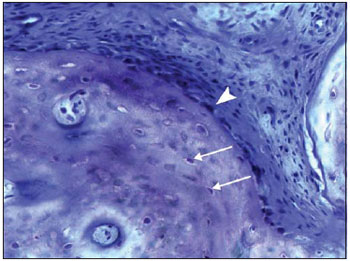
Picture 6. Histological cut of the tympanic bulla colored with toluidine blue (200x) showing a new osseous trabecula. See the presence of osteocytes inside the trabecula (white arrow) and osteoblasts in its periphery (arrow tip). Absence of neutrophils and multinuclear giant cells of foreign body type.
In our study, we confirmed that there were no clinical signs of rejection to the material such as the presence of granulomatous inflammatory reaction signs and implant extrusion signs. In the histopathological analysis, no samples that received the alpha-tricalcium-phosphate cement presented foreign body type inflammatory reaction signs, which normally occur in the material rejection processes.
This study also showed that the use of alpha-tricalcium-phosphate bone cement did not present complications when used to obliterate the guinea pig mastoid cavity. The absence of effusion in middle ear is a sign that there was the conservation of the middle ear physiology.
As for the ossification grade, we found significantly minor ossification in the cement group compared to the no-cement group (Picture 7). Such information confirms the studies that show this material is not osteoconductive. The alpha-tricalcium-phosphate bone cement is microporous, which does not allow the internal osseous growth (24). However, there were signs of reabsorption and osseous growth in the implant periphery (Picture 8), confirming that in time the material may be reabsorbed and replaced for new bone. Studies in animals show that the alpha-tricalcium-phosphate bone cement undergoes osseous remodeling similar to what occurs in the normal bone. Such material undergoes osteoclastic reabsorption followed of osteoblastic migration and formation of new bone (24). The properties of drying even in moist places and adherence to the bone may form the advantages compared to hydroxyapatite. The mastoid cavity is moist and has many times the presence of blood. In addition, the fact the material adheres to the adjacent bone is a factor that may represent a greater stability in the surgical bed. In all samples studied, the cement was firmly adhered inside the tympanic bulla.
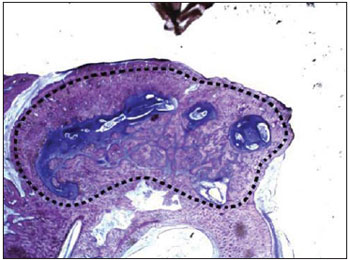
Picture 7. Histological cut of the tympanic bulla of the control group colored with toluidine blue (12.5x) showing the cavity intense ossification (dotted).
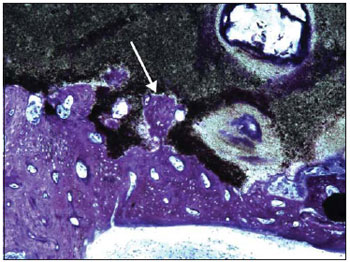
Picture 8. Histological cut of the tympanic bulla of cement group colored with toluidine blue (100x) showing a new osseous trabecula (arrow) invading the alpha-tricalcium-phosphate bone cement (black amorphous substance).
The alpha-tricalcium-phosphate bone cement is biocompatible in guinea pigs mastoid cavity.
The alpha-tricalcium-phosphate bone cement undergoes osseous remodeling, but it does not present osteoconduction property.
BIBLIOGRAPHICAL REFERENCES1. Bento RF, Miniti A, Marone SAM. Tratado de Otologia.1ª ed., São Paulo: EDUS; 1998.
2. Karmarkar S, Bhatia S, Saleh E, et al. Cholesteatoma surgery: the individualized technique. Ann Otol Rhinol Laryngol. 1995, 104:591-595.
3. Chang C, Chen M. Canal-wall-down tympanoplasty with mastoidectomy for advances cholesteatoma. J Otolaryngol. 2000, 29:270-273.
4. Roden D, Honrubia V, Wiet R. Outcome of residual cholesteatoma and hearing in mastoid surgery. J Otolaryngol. 1996, 25: 178-181
5. Black B. Mastoidectomy elimination. Laryngoscope. 1995, 105(12 Pt 2):1-30.
6. Mahoney JL. Tympanoacryloplasty. Arch Otolaryngol Head Neck Surg. 1962, 75:519-522.
7. Mahr MA, Bartley GB, Bite U, Clay RP, Kasperbauer JL, Holmes JM. Norian Craniofacial Repair System Bone Cement for the Repair of Craniofacial Skeletal Defects. Ophthalmic Plastic & Reconstructive Surgery. 2000, 16(5):393-398.
8. Baker S, Weinzweig, J, Kirschner RE, Bartlett SP. Applications of a New Carbonated Calcium Phosphate Bone Cement: Early Experience in Pediatric and Adult Craniofacial Reconstruction. Plastic & Reconstructive Surgery. 2002, 109(6):1789-1796.
9. Kirschner RE, Karmacharya J, Ong GBS, Gordon AD, Hunenko OBA, Losee J, Gannon FH, Bartlett SP. Repair of the Immature Craniofacial Skeleton With a Calcium Phosphate Cement: Quantitative Assessment of Craniofacial Growth. Annals of Plastic Surgery. 2002, 49(1):33-38.
10. Goksu N, Haziroglu R, Kemaloglu Y, Karademir N, Bayramoglu I, Akyildiz N. Anatomy of the guinea pig temporal bone. Ann Otol Rhinol Laryngol. 1992, 101(8):699-704.
11. Wysocki J. Topographical anatomy of the guinea pig temporal bone. Hear Res. 2005, 199(1-2):103-10.
12. Linthicum FH Jr.The fate of mastoid obliteration tissue: a histopathological study. Laryngoscope. 2002 , 112(10):1777-81.
13. Leatherman BD, Dornhoffer JL, Fan CY, Mukunyadzi P. Demineralized bone matrix as an alternative for mastoid obliteration and posterior canal wall reconstruction: results in an animal model. Otol Neurotol. 2001, 22(6):731-6.
14. Rosenblut B, Ahlvin RC, Carr CD, et al. Silicone implants in the mastoid portion of the temporal bone. Ann Rhinol Laryngo.l 1966, 75: 889- 89.
15. Shea JJ, Malenbaun BT, Moretz WH. Reconstruction of the posterior wall with proplast. Otolaryngol Head Neck Surg. 1984, 92:329-333.
16. Helms J, Geyer G. Closure of the petrous apex of the temporal bone with ionomeric cement following translabyrinthine removal of an acoustic neuroma. J Laryngol Otol. 1994, 108: 202-205.
17. Bryan DL, Dornhoffer JL. Bioactive Glass Ceramic Particles as an alternative for mastoid obliteration: results in a animal model. Otol & Neurotol. 2002, 23(5):657-660.
18. Jang CH, Cho YB, Bae CS. Evaluation of bioactive glass for mastoid obliteration: a guinea pig model. In Vivo. 2007, 21(4):651-5.
19. Costantino PD, Hiltzik D, Govindaraj S, Moche J. Bone healing and bone substitutes. Facial Plast Surg. 2002, 18(1):13-26.
20. Yung MW. The use of hydroxyapatite granules in mastoid obliteration. Clin Otolaryngol. 1996, 21:408-484.
21. Jahn AF. Experimental applications of porous (coralline) hydroxyapatite in middle and mastoid obliteration. Laryngoscope. 1992, 102:289-299.
22. Hussain A, Ram B, Hilmi OJ. Reconstruction of Mastoid Cavity With Hydroxyapatite Cement and Postauricular Flap. Laryngoscope. 2002, 112(3):583-585.
23. Frankenburg EP, Goldstein SA, Bauer TW, et al. Biomechanical and histological evaluation of a calcium phosphate cement. J Bone Joint Surg Am. 1998, 80:1112-1124.
24. Constantz BR, Ison IC, Fulmer MT, et al. Skeletal repair by in situ formation of the mineral phase of bone. Science 1995, 267:1796-1799.
25. Ison I, Fulmer B, Barr B, et al. Synthesis of Dahllite. The Mineral Phase of Bone. Boca Raton, FL: CRC Press Lewis Publishers; 1994.
26. Kopylov P, Jonsson K, Thorngren KG, et al. Injectable calcium phosphate in the treatment of distal radius fractures. J Hand Surg. [Br] 1996, 21:768-771
27. Kopylov P, Runnqvist K, Jonsson K, et al. Norian SRS versus external fixation in redisplaced distal radial fractures. A randomized study in 40 patients. Acta Orthop Scand. 1999, 70:1-5.
28. Hoffmann KC, Kuhn JJ, Strasnick. N. Bone Cements as Adjuvant Techniques for Ossicular Chain Reconstruction. Otol & Neurotol. 2003, 24: 24-28.
29. McDowell MP, Kuhn JJ, Strasnick B. Incudostapedial Joint Reconstruction Using Dahllite Bone Cement in a Chinchilla Model. Otolaryngol Head Neck Surg. 2005, 133(2) Supplement 1:P83-P84.
1. Doctor of Medicine by the Medicine College of USP. Assistant Doctor of the HCFMUSP's Otorhinolaryngology Department Otology Group.
2. Doctor of Nephrology by the Medicine College of USP. Assistant Doctor of the Nephrology Department of the Clinical Hospital of São Paulo.
3. Free Professor for the Medicine College of USP. Titular Professor of the Otorhinolaryngology Discipline of the Medicine College of USP.
4. Free Professor for the Medicine College of USP. Assistant Doctor of the Otorhinolaryngology Department of USP.
Institution: Hospital das Clínicas da Faculdade de Medicina da USP.
São Paulo / SP - Brazil
Mail address: Robinson Koji Tsuji
Alameda Ibérica 285, Casa 312 - Bairro Tamboré
Santana de Parnaíba / SP - Brasil - CEP 06543-502
E-mail: rktsuji@uol.com.br
FAPESP Financial support.
Article received on September 18, 2008.
Approved on September 30, 2008.