INTRODUCTIONAging is a complex, dynamic and progressive process, in which there are morphological, functional, biochemical and psychological modifications (1). This process is determined by various factors that are present since the birth, keep on developing along the years and have a large variation between individuals (2, 3, 4).
One of the aging variability factors are the habits harmful to health, amongst which, smoking, that may interfere or accelerate the development stage and is taken as one of the most critical public health problems of mankind (5). Today smoking causes one third of all deaths by cancer in many western countries (6). And most smokers are in the developed countries (5).
Smoking is highly aggressive to the vocal tract and is one of the main risk factors for cancer of larynx. It may lead to the vocal tract irritation, vocal cords edema, hoarseness, coughing, increase of secretion and infections (4). Moreover, the smokers more stuck to the addiction have more damages in the quality of life and present symptoms such as depression and anxiety (7).
The larynx tissues are affected by smoking (8). Larynx was taken as the organ most sensitive to histopathological changes after exposure to cigarette smoke. We found diffuse squamous metaplasia of the pseudostratified epithelium and squamous hyperplasia in the epiglottis base, depending on the dose (9).
Therefore, since smoking is harmful for the vocal cords and a few studies describe the macroscopic changes in such structures, it's crucial to investigate the possible morphological changes caused to the three dimensions of the vocal cords of elderly people with and without adhesion to this habit. The objective of this research is to evaluate the vocal cords dimensions in smoker and non-smoker elderly people of the male sex.
METHOD15 male corpses were part of the study, and they were 7 smokers and 8 non-smokers, in the age range from 60 to 90 years, from the Death Verification Service (DVS). It was an experimental study.
The cases selected met the inclusion and exclusion criteria. In the research, we included non-violent death victims with up to 24 post-mortem hours and aged over 60 years.
We excluded individuals younger that 60 years old previously submitted to tracheostomy, laryngeal surgery, radiotherapy in the cervico-facial region, affected by benignant or malignant neoplasms in the cervicothoracal region or any pathology that affected the laryngeal region.
For data collection, four sequential steps were followed:
1
st) The clinical history of the corpse: we made notes in the vocal cords morphometry protocol (Annex A), specifically prepared for the research, in order to document the corpse's general data and clinical history.
2
st) Larynx removal: after selection and completion of the protocol, during autopsy of the corpse we carried out the removal of the larynx through incision in the skin of the upper thoracic region and raising of the cervicothoracal cutaneous piece. The larynx was removed in block after section on the hyoid bone and to the level of the fourth tracheal ring.
3
rd) Larynx removal: the larynx was dissected carefully and the larynx extrinsic musculature, vessels and nerves were removed.
4
th) Vocal cards dimensions morphometry: the dissected larynxes were not fixed by any kind of substance, so as to prevent deformations to the tissue and changes to the measures.
The localization points between the dimensions of the vocal cords membranous portion were based on a morphometrical study of the vocal cords (10) and the measures carried out by the same researcher in order to avoid vices. The measures were carried out in millimeters with Stainless Hardened digital slide gauge of 0.01mm of accuracy.
The measures order was made in the following sequence: width, thickness and length at the right and left sides respectively.
The length was measured with the digital slide gauge depth rod with localization since the vocal cord insertion in the thyroid cartilage, below the epiglottis petiole, up to its insertion in the vocal process of the arytenoid cartilage.
The width was measured with the digital slide gauge external rod with localization set forth with the distance comprised between the free edge and the transition between the vocal linking and the laryngeal ventricle floor.
The thickness was measured with the digital slide gauge external rod with localization defined as the distance between two parallel plans draw at the upper or lower part of the thyroarytenoid muscle (TA).
The data collected were organized in the Microsoft Excel 2003 and distributed in the form of tables and graphic for a better view. For analysis of the data we applied parametric tests such as the t of Student in the comparisons as for the vocal cords dimensions and smoking. We considered the significance index of 95% (p<0.05). The statistical program used was BioStat version 5.0. For ethical considerations this research was approved by the Ethics Committee in Research with Human Beings with register no. 309/07.
RESULTSIn Graphic 1, we observed the average comparison of the vocal cords length of elder smokers, that was of 15.61mm and of non-smokers of 16.16mm. In the statistical calculation through test t Student (p<0.05), the significance index was of p=0.58, and there was no significant statistical difference between both groups. Thus, the length was not changed by the smoking effect.
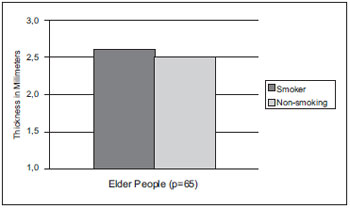
Graphic 1. Vocal cords length average comparison in smoker and non-smoker elder people of the male sex.
In Graphic 2, we may view the vocal cords length measures comparison between the smoking elders group with an average of 2.37mm and non-smokers group of 2.36mm, in which the significance index was of p=0.72; therefore there was no significant difference in this measurement in the groups studied. The width between smokers and non-smokers was equivalent.
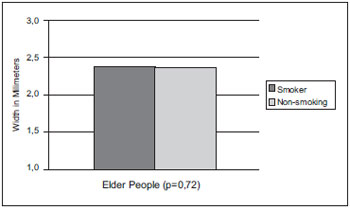
Graphic 2. Vocal cords width average comparison in smoker and non-smoker elder people of the male sex.
As for the vocal cords thickness measurement in the elders, the smoking group average was of 2.66mm and that of non-smokers was of 2.50mm. We may identify in Graphic 3 that there was no significant statistical difference between the groups (p=0.65). However, the average shows a thickness minimally higher in the smoking group.
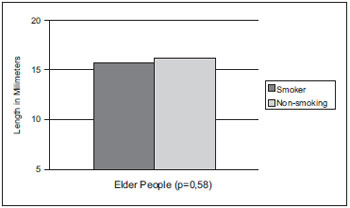
Graphic 3. Vocal cords thickness average comparison in smoker and non-smoker elder people of the male sex.
Today 1.3 billion of the world population is of smokers. Out of whom, approximately one billion is of the male sex.
In the elderly smokers group, the vocal cords length ranged from 13.27mm-18.54mm with an average of 15.61mm; in the non-smokers group it ranged from 13.62mm-17.81mm with an average of 16.16mm. In the adult man the membranous portion of the vocal cords ranges from 11.5mm to 16mm (11). According to Graphic 1, which describes the comparison of the vocal cords length average of smoker and non-smoker elders, there was no significant change in the length.
The literature shows (11) that the longer the vocal cords length is, the lower is the fundamental frequency, which makes the voice graver. In the elderly people, other studies indicate an increase of the fundamental frequency in non-smoking man and a decrease in women (12) due to the aging process that causes anatomic and physiologic changes in the larynx and vocal cords (13). According to some authors, one of the anatomic changes is the shortening of the vocal cords length that occurs with aging and was observed through histological study (3).
Some authors confirmed (14) a significant difference between the fundamental frequency of smoking and non-smoking adult men in the oral reading and in the spontaneous speech. The smokers had a lower fundamental frequency than the non-smoking values and in the women the trend was the same.
In a longitudinal study (15) with 11 men aged from 50 to 81 years during 5 years, male smokers had a lower fundamental frequency than non-smokers of the same sex, but this seemed to be reversible for those who would have quit smoking. We assume this capacity of reversibility is due to the diminishment of the edema caused by the smoke (4), and makes the voice less grave after ceasing smoking. Other authors (16) researched the fundamental frequency before, during and after a period of 40 hours without smoking and confirmed the smoking effects may be reverted after 40 hours of smoking ceasing. Despite the measurements were made with up to 24 hours post-mortem, the information granted by the relatives on the time of smoking of the researched individuals was many times imprecise, which may have influenced our findings.
As for the passive smoking, studies (17) showed that the vocal cord function and structure were not changed negatively by this exposure. However, another research carried out with (18) rats exposed to cigarettes confirmed there was hyperplasia and squamous metaplasia in the free extremity of the vocal cords and squamous hyperplasia in the vocal cord medial portion and concluded the passive inhaling of cigarette smoke caused critical morphological changes in the vocal cord epithelium and could progress into neoplasm.
In our study, the possible histological alterations in the vocal cords structure were not evaluated, because the objective was focused on the macroscopic aspect, considering the absence of studies in this matter.
Researchers (19) evaluated the effect of smoking on the voice of 134 young smokers and non-smoker with short time of smoking and suggested a possible neurological effect of nicotine or some other chemical component of the tobacco due to the fundamental frequency disturbance in both sexes, mainly in women and the vocal trembling present in men.
Graphic 2 presents the width average comparison in elderly smokers and non-smokers of the male sex. The smoking provokes edema (4) in the vocal cords and reduction of the fundamental frequency (16). Reinke's Edema is a diffuse lesion of the vocal cords characterized by the increase of the vocal cord mass by fluid that leads to a grave pitch (20) and is associated to the consumption of tobacco, that is, its occurrences in smokers is high. A research (21) showed that the this disease clinical manifestation relates to the number of cigarettes smoked per day, that is, the longer the duration of the exposure to the cigarette smoke, the higher the level of histological harm. Therefore, it was suggested the width dimension could be committed in some degree in smoking individuals; however, by the macroscopic morphometry there was no statistically significant difference in the results.
As for the vocal cords thickness dimension of smoking and non-smoking elders, in Graphic 3 we viewed there was no significant difference between the groups. However, studies (8) show there is a thickness increase of the smokers and etilists vocal cords epithelium, when compared to the same tissue of non-smoker and non-etilists. Therefore, the smoking-alcohol association may make morphological changes potential, but the differences found must be limited to the microscopic level.
The cigarette smoke represents an important accelerator of the aging process, whether directly through complex mechanisms predominantly measured by the excessive formation of free radicals, or indirectly, which favors the appearing of several pathologies having the smoking as a risk factor, such as: cancer, cardiovascular and respiratory diseases (22).
Today, at least, 15% of the total cancer cases are ascribable to smoking. The tobacco products, among the most consumed ones, the cigarettes, contain more than 50 carcinogenic established or identified substances and these may increase the risk of cancer for causing mutations that disturb the cellular cycle regulation, or through their effects on the immune or endocrinal system. Some factors like genes, the diet and environmental exposures may change the smokers cancer susceptibility (23).
The association between smoking and cancer is already broadcast. In addition to cancers already proved to be associated to smoking, such as larynx, lung, oral cavity, pharynx, esophagus, pancreas, bladder and renal pelvis, some studies show the tendencies to other cases, such as nasal cavity and paranasal sinuses cancer, nasopharynx, stomach, liver, kidneys (renal cells carcinoma), mouth of uterus, esophagus adenocarcinoma and myeloid leukemia (6). Due to this fact, non-smokers seem to be able to retard the appearing of diseases and the aging process, and reach longevity (22).
The complexity of the aging process and the method used are responsible for the variability of scientific findings in the elderly population.
We suggest studies with a larger sample, and, if possible, associated to more details of the clinical history regarding smoking such as time, type of smoke, quantity and association to alcohol.
CONCLUSIONAccording to the results obtained, we may conclude that by means of the vocal cords macroscopy morphometry we found no significant differences between the dimensions of length, width and thickness in smoking and non-smoking male elderly people.
BIBLIOGRAPHICAL REFERENCES1. Netto MP. O estudo da velhice no século XX: histórico, definição do campo e termos básicos. Em: Freitas EV et al. (Org.) Tratado de geriatria e gerontologia. Rio de Janeiro: Guanabara Koogan; 2002, pp. 2-12.
2. Brasolotto AG. Voz na Terceira Idade. Em: Ferreira LP, Befi-Lopes DM, Limongi SCO. Tratado de Fonoaudiologia. São Paulo: Roca; 2004.
3. Hirano M, Kurita S, Sakaguchi S. Ageing of the Vibratory Tissue of Human Vocal Folds. Acta Oto-Laryngologica. 1989, 107(5 e 6):428-33.
4. Pinho SMR. Avaliação e Tratamento da Voz. Fundamentos em Fonoaudiologia. Rio de Janeiro: Guanabara Koogan; 1998, pp. 20-2.
5. Menezes AMB. Diretrizes para Cessação do Tabagismo. Epidemiologia do Tabagismo. J Bras Pneumol. 2004, 30(Supl2).
6. Sasco AJ, Secretan MB, Straif K. Tobacco smoking and cancer: a brief review of recent epidemiological evidence. Lung Cancer. 2004, 45(Suppl.2):S3-S9.
7. Castro MG, Oliveira MS, Moraes JFD, Miguel AC, Araujo RB. Qualidade de vida e gravidade da dependência de tabaco. Rev. Psiq. Clín. 2007, 34(2):61-7.
8. Hirabayashi H, Koshii K, Uno K, Ohgaki H, Nakasone Y, Fujisawa T et al.Laryngeal epithelial changes on effects of smoking and drinking. Auris Nasus Larynx. 1990, 17(2):105-14.
9. Haussmann H, Anskeit E, Becker D, Kuhl P, Stinn W, Teredesai A et al. Comparison of fresh and room-aged cigarette sidestream smoke in a subchronic inhalation study on rats. Toxicol Sci. 1998, 41: 100-16.
10. Ximenes Filho JA. Melo ECM. Carneiro CG. Tsuji DH. Sennes LU. Correlação entre a altura e as dimensões das pregas vocais. Rev Bras Otorrinolaringol. 2003, 69(3):371-74.
11. Behlau M. Voz - O Livro do Especialista. Rio de Janeiro: Revinter; 2001, pp 309-12.
12. Bressan VL. Influências no envelhecimento da voz. Curitiba, 1999, (Monografia - Centro de Especialização em Fonoaudiologia Clínica).
13. Martins RHG. A Voz e seus distúrbios. São Paulo: Cultura Acadêmica; 2005.
14. Sorensen D, Horii Y. Cigarette smoking and voice fundamental frequency. J Commun Disord. 1982, 15 (2):135-44.
15. Verdonck-de Leeuw IM, Mahieu HF. Vocal aging and the impact on daily life: a longitudinal study. J Voice. 2001, 18(2):193-202.
16. Murphy CH, Doyle PC. The effects of cigarette smoking on voice-fundamental frequency. Otolaryngol Head Neck Surg. 1987, 97(4):376-80.
17. Lee L. Stemple JC. Geiger D. Goldwasser R. Effects of Environmental Tobacco Smoke on Objective Measures of Voice Production. Laryngoscope. 1999, 109(9):1531-4.
18. Duarte JL, Faria FAC, Ceolin DS, Cestari TM, Assis GF. Efeitos da inalação passiva da fumaça de cigarro sobre as pregas vocais de ratos. Rev Bras Otorrinolaringol. 2006, 72 (2):210-6.
19. Gonzalez J, Carpi A. Early effects of smoking on the voice: A multidimensional study. Med Sci Monit. 2004, 10(12):649-56.
20. Matsuo K, Kamimura M, Hirano M. Polypoid vocal folds: a ten years review of 191 patients. Auris Nasus Larynx. 1983, 10(Suppl.):37-45.
21. Marcotullio D, Magliulo G, Pezone T. Reinkes edema and risk factors: clinical and histopathologic aspects. Am J Otolaryngol. 2002, 23(2):81-4.
22. Nicita-Mauro V, Lo Balbo C, Mento A, Nicita-Mauro C, Maltese G, Basile G. Smoking, aging and the centenarians. Exp Gerontol. 2008, 43(2):95-101.
23. Kuper H, Adami H-O, Boffetta P. Tobacco use, cancer causation and public health impact. J Intern Med. 2002, 251:455-66.
1. Master's Degree in Pathology and Applied Morphology at UFPE - Specialization in Language with focus on Neurosciences. Phonoaudiologist.
2. Doctor's Degree in Medicine at UFPE. Affiliate Professor I at UFPE.
3. Doctor's Degree in Nutrition at UFPE. Assistant Professor I at UFPE.
4. Master's Degree in Pathology - Applied Morphology at UFPE. Phonoaudiologist.
Institution: Departamento de Patologia da Universidade Federal de Pernambuco. Vitória de Santo Antão / PE - Brazil.
Mail address:
Sandrelli Virginio de Vasconcelos
Rua 01, Nº 115 - Bela Vista - Vitória de Santo Antão
PE - Brazil - Zip Code: 55608-430
Phone nos.: (+55 81) 3523-1245 / 9402-4881 / 3235-3085
E-mail: sandrelli@hotmail.com
Article received on November 26, 2008.
Approved on February 11, 2009.