INTRODUCTIONThe human BCL2 gene is located on chromosome 18q21 in a telomere-centromere orientation and consists of three exons (Figure 1). The BCL2 protein found in the inner mitochondrial membrane regulates apoptosis by inhibiting the cells from programmed cell death (1). Translocation (14; 18) (q32, q21), detected in 70-85% of follicular lymphomas, leads to overexpression of BCL2 protein by the juxtaposition of the BCL2 gene to JH gene segments of immunoglobulin heavy chain (IGH). Most of the points of breaks of t (14; 18) occur in non-coding region of 18q21.3 BCL2. These points are the regions of breaks MBR (Major Breakpoint Region), found in approximately 60% of lymphomas with t (14; 18) and mcr (Minor Cluster Region), and located 20 kb away from the MBR region (2.3, 4.5).
Data from recent studies are contradictory on the correlation between the presence of rearrangements and changes in gene expression as predictive values of BCL2/JH prognosis of follicular lymphomas (RADOJKOVIE et al., 2008) (6). However, for DEGHIED et al. (2007) (7) the expression of this protein may be used, including retrospectively for the diagnosis of doubtful cases, such as reactive hyperplasia. In patients with minimal expressions BCL2/JH in samples of peripheral blood or bone marrow would be the correct retest the rearrangement BCL2/JH to exclude false positives (8,9).
Unlike the findings in lymphomas, the over expression of BCL2 protein has been detected in 30% of carcinomas of the head and neck (10,11,12) and is not associated with presence of the t (14; 18) (HARN et al., 1996). Few epidemiological studies have indicated that the use of tobacco and high consumption of alcohol are important etiologic factors in the induction of genetic alterations such as chromosomal translocation t (13, 14, 21). However, the rearrangement BCL2/JH has been found in lymphocytes from healthy individuals who smoke (14,15,16,17,18) and in bone marrow cells from patients with diseases other than lymphoid (8,17).
The aim of this study was to investigate the presence of BCL2 rearrangement (MBR) / JH in tissue samples from patients with carcinomas of the mouth and pharynx and the possible correlation with the same exposure to tobacco and alcohol.
METHODGenomic DNA was extracted from 16 fresh biopsies of cancers of the mouth (n = 13) and pharynx (n = 3) obtained from patients with histological confirmed epidermoid carcinoma, maid to any treatment by digestion of tumor tissue by proteinase-K (20 mg / ml) and DNA extraction without phenol-chloroform. The tumor biopsies were performed in the service of Otolaryngology School of Medicine, UNESP, Botucatu, SP, Brazil, after the patient and or guardian becomes aware of the research objectives and signed a consent form and approved by the Ethics in Research in humans of our institution.
The presence of rearrangement BCL2/JH (Figure 1), breaking MBR region was investigated by nested PCR. 1.0
mg of DNA from each sample was amplified with 200 nmol of MBR and JH primers (GRIBBEN et al., 1994) in 50
ml of buffer (Tris-HCl pH 7.5 and 5 mM, 25 mM KCl MgCl2 and 1.5 mM), 0.1 mM dNTPs and 1.25 U Taq DNA polymerase. This was followed by 25 amplification cycles at 940C for 1 minute, 550C for 1 minute and 720C for 1 minutes. In the second reaction (nested) 10
ml of the product's first reaction was amplificated again with primers MBR-JH-N and N (200 nmol) in 50
ml of buffer (Tris-HCl pH 7.5 to 8 mm, the KCl 40 mM MgCl2 and 1.5 mM), 0.1 mM dNTPs and 1.25 U Taq DNA polymerase. This was followed by 30 cycles of amplification at 940C for 1 minute, 580C for 1 minute and 720C for 1 minute. The products of nested PCR were subjected to electrophoresis on polyacrylamide gel and stained with 5% solution of ethidium bromide (50 mg / ml).
DNA extracted from cell line RL with the MBR rearrangement was used as positive control of PCR reaction. Human DNA negative at (14, 18) and a tube of PCR without DNA were used as negative controls of the reaction.
The Fisher exact test was used to assess the correlation between the presence of BCL2 rearrangement (MBR) / JH and exposure to tobacco and alcohol.
RESULTSThe incidence of BCL2 rearrangement (MBR) / JH was 12.5% (2 / 16) for cases of epidermoid carcinoma of the mouth and pharynx (Table 1).
The analysis of the frequency of BCL2 rearrangement (MBR) / JH was made by molecule (
l) in the two positive patients (Figures 2, 3 and 4) and used a Poisson model of statistical analysis for the number of rearrangements per sample (21). Because all trials had the same number of molecules (1
mg of DNA contains 5 x 10-19 moles of each single copy sequence = ~ 300 000 molecules), the estimate
l was given by the usual Poisson estimator:
l= 1 / M . ln (1 - p), where p is the fraction of trials with at least one rearrangement.
No association was found between the presence of BCL2 rearrangement (MBR) / JH and exposure to tobacco and alcohol (Table 2) (P = 0.6545).
PCR products of patients positive for the rearrangement BCL2/JH can be observed in Figure 2. Fragments of different sizes were observed in two patients with squamous cell carcinoma.
Figures 2, 3 and 4 show the amplifications carried out on multiple screens, rearrangement of BCL2 (MBR) / JH of the two patients who were positive.
The second patient, a smoker since childhood, and chronic alcoholics (~ two liters of alcohol per day), remained positive in 3 of 8 more screens performed (Figure 2A). Were observed fragments of the same size, suggesting that it is the same clone. The frequency of the MBR rearrangement was 19.32 x 10 -7 molecules positive.
Patient 6, chronic smoker and drinker, he remained positive in seven of eight more trials performed (Figure 2B). Fragments were observed even sizes (~ 200 bp). The frequency of the MBR rearrangement was 73.57 x 10 -7 molecules positive.
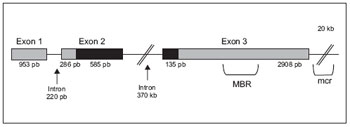
Figure 1. BCL2 gene structure. The BCL2 contains three exons and two introns. The untranslated regions of the gene are illustrated with solid bars of gray, while black bars represent the translated regions.
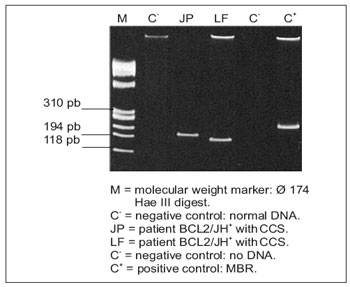
Figure 2. Amplifications of BCL2 rearrangement (MBR) / JH in patients with squamous cell carcinoma (EC). Polyacrylamide gel electrophoresis in 5%.
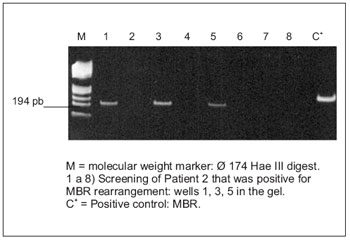
Figure 2A. Amplifications multiple BCL2 rearrangement (MBR) / JH in patients with squamous cell carcinoma (EC). Polyacrylamide gel electrophoresis in 5%.
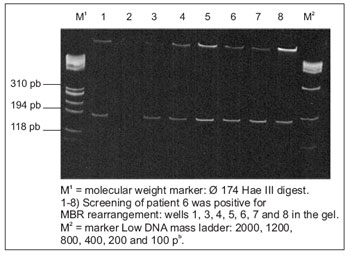
Figure 2B. Amplifications multiple BCL2 rearrangement (MBR) / JH in patients with squamous cell carcinoma (EC). Polyacrylamide gel electrophoresis in 5%.
A high expression of BCL2 has been observed in squamous cell carcinomas (LAVIEILLE et al. 1998; DRENNING et al., 1998), however, there are reports of the presence of BCL2/JH rearrangement in tumors of head and neck. In this study, the BCL2 rearrangement (MBR) / JH was investigated in fresh biopsies of 13 squamous cell carcinomas and 3 carcinomas, from patients with head and neck tumors by nested PCR.
HARN et al. (1996) (12) reported the absence of the MBR rearrangement by analyzing 32 cases of nasopharyngeal carcinoma by means of PCR. However, the study by HARN et al. (1996) (12) were used for fixed tissue samples. Thus, this discrepancy in the incidence of BCL2/JH rearrangement may be due to technical differences or sampling.
SALO et al. (1997) (20) studied the status of the BCL2 gene in nine human cell lines of squamous cell carcinoma. The BCL2 cDNA was amplified in five cell lines, showing that the mRNA was expressed in these cells. The five cDNAs were sequenced; however, point mutations in the BCL2 gene were not detected, indicating that translocation is probably a typical change in these neoplastic cells. For VERGIES et al. (2004) (18) these results suggest that one mechanism of BCL2 overexpression in squamous cell carcinomas, may be due to chromosomal translocation t (14;18).
BELL et al. (1995) (13) and SHULER et al. (2003) (17) reported a significant association between smoking and the frequency of cells with at (14, 18) in peripheral blood of healthy volunteers.Other authors have found these same translocations in individuals with cancer are associated with tobacco (8,9,15,16). In this study, there was an association between positive MBR/BCL2 rearrangement and exposure to tobacco and alcohol in patients with head and neck tumors. Because BCL2 mutation rate was similar for both smokers and nonsmokers (8.9 15), the relationship of tobacco antigens stimulating the emergence of clones of rare rearrangements BCL2/JH pre-existing was not observed in our patients. However, no such association was impaired by the low number of subjects studied.
Still, in this study, one of the smokers showed a positive high frequency (73.57 x 10 -7) of BCL2 rearrangement (MBR) JH and this increase may be due to the expansion of a single dominant clone and sequencing of the product PCR can confirm that this individual observation. This data could have predictive value for diagnosis as shown by BELL et al. (1995), when one of the smokers (three packs of cigarettes a day), which showed a high frequency of rearrangement in peripheral blood BCL2/JH developed melanoma (21). This individual was positive four times in four trials conducted and remained positive on April 2 more screens retested eight months later. Thus, measuring the rate of translocation t (14, 18) could identify individuals with a greater risk of developing lymphoma or other cancers or that responded poorly to therapy (6,7,21).
Finally, one can say that the BCL2 rearrangement (MBR) JH is not restricted to lymphoproliferative diseases and can be detected in neoplastic cells of primary carcinomas of the mouth and pharynx. The translocation t (14; 18) may therefore be a secondary mutation found in squamous cell carcinoma of the mouth and pharynx but in our case, due to the small number of patients tested, cannot assess the prognostic value of the presence of BCL2 rearrangement / JH in these tumors.
BIBLIOGRAPHICAL REFERENCES1. Eckenrode EF, Yang J, Velmurugan GV, Foskett JK, White C. Apoptosis protection by Mcl-1 and Bcl-2 modulation of inositol 1,4,5-Trisphosphate receptor-dependent Ca2+ signaling. J. Bid. Chem. 2010, 285:285-97.
2. Lee MS. Molecular aspects of chromosomal translocation t(14;18). Seminars in Hematology. 1993, 30:297-305.
3. Heim S, Miltelman F. Malignant Lymphomas. (1995). In: Cancer Cytogenetics (Heim S, and Miltelman F, eds.). New York: Wiley-Liss, pp. 266-309.
4. Gaidano G, Dalla-Favera R. (1997). Molecular Biology of lymphomas. In: Cancer: Principles and Practice of Oncology (Devita Jr VT, Hellman S, and Rosenberg S, eds.). Philadelphia: J. B. Lippincott Company, pp.2131-2145.
5. Monni O, Franssila K, Joensuu H, Knuutila S. BCL2 overexpression in diffuse large B-cell lymphoma. Leukemia and Lymphoma. 1999, 34:45-52.
6. Radojkovic M, Ristic S, Colovic M, Cemerikic-Martinovic V, Radojkovic D, Krtilica K. Molecular characteristics and prognostic significance of Bcl-2/IgH gene rearrangement in Serbian follicular lumphoma patients. Neoplasma. 2008, 55(5):421-7.
7. Deghedy H, Fonda M, Sharin D, Shamaa S, El-Bedewy A, Ghaffar HA. Diagnostic and prognostic utility of t(14;18) in follicular lymphoma. Acta Haematologica. 2007, 118(4):231-6.
8. Bownan A, Jones D, Medeiros J, Luthra R. Quantitative PCR detection of t(14;18) Bcl2/JH. Fusion sequences in follicular lymphoma patients. JMD. 2004, 6(4):129-36.
9. Karubec K, Guo Y, Suzumiya J et al. CD10(-) MUM1(+) follicular lymphomas lacks BCL2 translocation and clinical features. Blood. 2007, 109:3076-9.
10. Lavieille JP, Gazzeri S, Riva C, Reyt E, Brambilla C, Brambilla E. p53 mutations and p53, Walf-1, Bax and Bcl-2 expression in field cancerization of the head and neck. t(14;18). Anticancer Res. 1998, 18:4741-9.
11. Drenning SD, Marcovitch AJ, Johnson DE, Melhem MF, Tweardy DJ, Grandis JR. Bcl-2 but not Bax expression is associated with apoptosis in normal and transformed squamous epithelium. Clin Cancer Res. 1998, 11:2913-21.
12. Harn HJ, Ho LI, Liu CA, Liu GC, Lin FG, Lin JJ, Chang JY, Lee WH. Down regulation of bcl-2 by p53 in nasopharyngeal carcinoma and lack of detection of its specific t(14;18) chromosomal translocation in fixed tissues. Histopathology. 1996, 28:317-23.
13. Bell DA, Liu Y, Cortopassi GA.Occurrence of bcl-2 oncogene translocation with increased frequency in the peripheral blood of heavy smokers. J Natl Cancer Inst. 1995, 87:223-4.
14. Hirose Y, Masaki Y, Ozaki M. Fluorescence in situ hybridization of chromossome FgH/BCL2 trabslocations for paraffin-embedded tissue: evaluation in follicular lymphomas. Int J Hematol. 2003, 78:154-9.
15. Schantz SP, Harrison LB, Forastiere AA. (1997). Tumors of the nasal cavity and paranasal sinuses, nasopharynnx, oral cavity, and oropharynx. In: Cancer: Principles and Practice of Oncology (Devita Jr VT, Hellman S. and Rosenberg S, eds.). Philadelphia: J. B. Lippincott Company, pp.741-801.
16. Xu J, Gimenez-Conti IB, Cunnigham JE, Collet AM, Luna MA, Lanfranchi HE, Spitz MR, Conti CJ. Alterations of p53, cyclin D1, Rb, and H-ras in human oral carcinomas related to tobacco use. Cancer. 1998, 83:204-12.
17. Schuler F, Hirt C, Dolken G. Chromossomal translocation t(14;18) in health individuals. Semim Cancer Biol. 2003, 13:203-9.
18. Vergier B, Beland-Rotureau MA, Benany MN, Beylot-Barry M, Dubus P, Delaunay M, Garroste JC, Taine L, Merlio JP. Neoplastic cells do not carry bcl2-JH rearrangements detected in a sulset of primary cutaneous follicle center B-cell lymphomas. Am J Pathol. 2004, 28(6):748-5.
19. Rauzy O, Galoin S, Chale JJ, Adoue D, Albarede JL, Delsol G, Saati TAL. Detection of t(14;18) carrying cells in bone marrow and peripheral blood from patients affected by non-lymphoid diseases. J Clin Pathol: Mol Pathol. 1998, 51:333-8.
20. Salo A, Servomaa K, Kiuru A, Pulkkinen J, Grénman R, Pekkola-Heino K, Rytomaa T. The bcl-2 gene status of human head and neck cancer cell lines. Acta Otolarynngol. 1997, 529:233-6.
21. Fuscoe JC, Setzer RW, Collard DD, Moore MM. Quantification of t (14;18) in the lymphocytes of healthy adult humans as a possible biomarker for environmental exposures to carcinogens. Carcinogenesis. 1996, 17:1013-20.
1 Full Professor in Otorhinolaryngology. Full Professor, Adjunct.
2 Doctoral and Postdoctoral Fellow in Genetics at UNESP.
3 PhD. Professor, Department of Medicine, Blood Bank Division.
Institution: Faculdade de Medicina de Botucatu - UNESP Department of Ophthalmology and Otolaryngology and Head and Neck. Botucatu / SP - Brazil. Mail Address: Jair Cortez Montovani - Faculty of Medicine of Botucatu - UNESP Department of Ophthalmology and Otolaryngology and Head and Neck District Rubiao Junior s / n - Botucatu / SP - Brazil - Zip code: 18603-970 - Telephone / Fax: (+55 14) 3811-6256 / 3811-6081 - E-mail: montovan@fmb.unesp.br
Article received in December 16, 2009. Article accepted in May 15, 2010.