INTRODUCTIONThe lexical development is directly connected to the language development. The performance of standardized tests of vocabulary has been used in many studies as a predictor of linguistic deficits both for normal hearing and hearing impaired children using hearing aid (AASI) and cochlear implant (CIC) (1, 2, 3, 4, 5, 6).
The MacArthur-Bates Communicative Development (7, 8, 9) and the Language Development Survey (LDS) (10, 11) are instruments largely used to check the lexical development. Both tests are composed by a checklist of words that must be completed by the parents. Concordant validations between both tests have already been proved and are widely accepted in the literature (11, 12). An LDS version and adaptation into Brazilian Portuguese has been published by Capovilla (13) and the name of the test was translated into List of Expressive Vocabulary Evaluation (LAVE). No researches have been reported with such instruments in Brazilian children using CI so far.
Researches reveal children with severe hearing loss have a deficit in the vocabulary repertoire compared to the normal-hearing children of the same chronological age (3, 14, 15, 16). One of the factors deemed to be critical is the fact the hearing system anatomic physiological integrity corresponds to a pre-requirement for the acquisition and normal development of the linguistic capabilities that consequently leads to a deficit in the lexical development. Another fundamental factor during the process of language development is the family intervention. The language development of children is positively influenced by the family involvement and mother and child interaction, and, consequently, the linguistic input provided by the family, which will determine the child's lexicon.
Based on the rehabilitation programs with participative family - a practice based on evidences - the objective of the research performed was the proposal of a program controlled for guidance to parents of deaf children who use cochlear implant. The program received the heading Participative Family for Deaf Children Using Cochlear Implant. This program approached topics connected to the development of linguistic and auditory capacities. The hypothesis of the study is the review of the program effectiveness by the children's performance variation, before and after the program, by the LAVE test.
METHODThis research was approved by the ethics committee of the institution under the protocol number 0290/10. All parents or responsible persons signed the free and clear authorization and agreed with the participation in the research and the further data publication. By following relevant ethic and moral precepts, the children's relatives in a control group were aware that in the case of confirmation of the study hypothesis there would be the prompt commencement of the guidance program specific for this group.
Casuistry
Eight deaf children using cochlear implant (CIC) attended in the Otorhinolaryngology Service of the Clinical Hospital of the Medical School of the University of São Paulo (HCFMUSP), as well as their respective families took part in the research.
The initial selection of the families was made based on the economic questionnaire Brazilian Economic Classification Criterion (17) (CCEB). This questionnaire classified the children into similar economic classes and level of education.
The participants were divided into two groups:
- Research Group (GI): four CIC and their respective parents, participants of the guidance program - CIC Participative Family;
- Control Group (GII): four CIC and their respective parents not integrating the guidance program.
The choice for composition of GI and GII was made by means of random numbers (Random Numbers Table Summary). The measurement of the groups was carried out based on the questionnaire of Family Permeability that enables to qualify the openness of the family as regards to the therapeutic process (18). The measurement criterion was by similar scoring or variation lower to 1.
Procedures
Step 1: Groups Composition
The parents (father and/or mother) were invited to avail their socioeconomic data voluntarily for the research. After agreement, the parents were invited individually in a separate room for completion of the questionnaire. During the completion of the questionnaire, the researcher presented the questions orally to the participants and transcribed their answers. The time used for completion ranged from five to ten minutes per participant. Once the economic classes and education levels were compatibilized, the groups were assorted and compared.
Step 2: Application of LAVE
The LAVE (pass/fail test in which the parents mark the words that are spontaneously spoken by the children in the several lexical categories) was applied in two collections. The pre-test was applied in GI and GII in the same period of time. The GI pre-test was applied immediately after the end of the program to all participants (between 30 to 40 days). The GII post-test was applied between 30 and 40 days after the pre-test to all participants.
Step 3: Achieving of the Participative Family Program for Deaf Children Using Cochlear Implant.
The Participative Family Program for Death Children Using Cochlear Implant was composed by four sessions, of weekly application, with duration of 60 minutes each. The duration of the program was of four weeks. The modules approached were: auditory and linguistic abilities. The sessions were structured as follows: objective; programmatic content; approaching technique and dialectical relation (19).
RESULTSIntra-group Analysis
The intra-groups analysis was carried out by considering the prevalence between the lexical categories in the pre-test and post-test and in the comparison before and after application of LAVE. We used the tests of Kruskal-Wallis ANOVA tests by rankings in the punctual testing and Friedman ANOVA for comparative testing. The Figures 1 and 2 allow the viewing of these results.
For 9GI we had the following:
- Pre-test - H (13. N=42)=22,007 p = ,055; indicating a tendency for differentiated performance between the categories;
- Post-test - H (13. N=42)=15,621 p = .270; indicating there is no differentiated performance between the categories;
- Pre-test x Post-test (N=42, gl=1) = 9.783 p = ,002*; indicating there is significantly differentiated outcome between pre-test and post-test performances.
For GII we had the following:
- Pre-test - H (13. N=42)=30,764 p = .003*; indicating a tendency for differentiated performance between the categories;
- Post-test - H (13. N=42)=15,459 p = 0.280; indicating there is no differentiated performance between the categories;
- Pre-test x Post-test (N=42, gl=1) = 2.454 p = ,117; indicating there is no significantly differentiated outcome between pre-test and post-test performances.
Analysis between Groups
The analysis between groups was carried out considering the performance between the groups in the pre-test, post-test and according to the lexical evolution of the groups. The test used was a one way ANOVA. Table 2 and Figure 3 allow the viewing of this comparison.
Comparison in the Pre-test - F(1.82)=1,9568,p=,1656, indicating there is no significant difference between the groups.
Comparison in the Post-test - F(1.82)=,0006,p=,979, indicating there is no significant difference between the groups.
Pre-test x Post-test Comparison - In spite the mean evolution in the lexical performance was higher for GI, there's no statistically significant difference between both groups through one way ANOVA.
DISCUSSIONThe quantitative analyses of the study appoint there is no influence from the CIC Participative Family on expansion of the vocabulary when the LAVE test is used (family perception).
In a qualitative analysis we may verify for Group I the average of words acquired was higher than for GII. For each word acquired by Group II, 2,18 words were acquired by Group I indicating a positive influence of the CIC Participative Family program on the children's vocabulary expansion.
Another qualitative detail was observed during application of the program - the learning of the families as regards to the linguistic and auditory abilities of their children.
The design of the research may be characterized as experimental and the data obtained is taken as a pilot study on the subject matter. In the expansion of the study both the program extension and the introduction of an objective instrument of evaluation of the children's lexicon development must be taken into account. The number of participants is an important variable and should be expanded, although it is compatible with similar studies.
Despite the limitations of the study this research is critical faced with the limited bibliographical reference about the theme - language development in Brazilian children using Cochlear Implant. With evolution of the Brazilian studies in this area the cross-language comparison will be possible and consequently the knowledge on the actual influence of the country's language in the language development in of the children using cochlear implant.
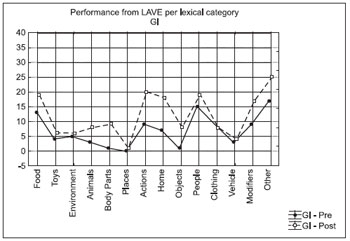
Figure 1. Performance from LAVE per lexical category - GI.
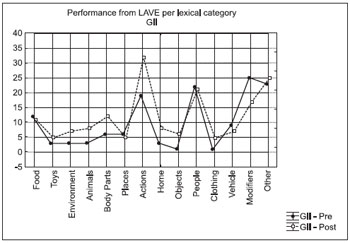
Figure 2. Performance from LAVE per lexical category - GII.
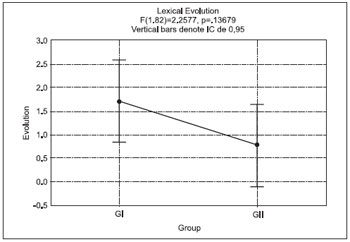
Figure 3. Lexical Evolution of both groups (GI and GII).
The hypothesis of the study was not confirmed. The groups do not differ is the review of the program effectiveness by the children's performance variation, before and after the program, by the LAVE test. This result must be taken as an important indicator of the vocabulary evolution evaluations based exclusively on the family perception. The main variables that should be considered in the future studies are: expansion of the sample; objective testing with validated vocabulary evaluation instrument; the program temporal extension.
BIBLIOGRAPHICAL REFERENCES1. Torres MLGM, Maia HA, Perissinoto J, Ferreira VJA. Descrição do Léxico Expressivo de Crianças aos 5 anos de idade. Rev Cefac. 2002, 4:241-251.
2. Nott P, Cowan R, Brown M, Wigglesworth G. Early Language Development in Children with Profound Hearing Loss Fitted with a Device at a Young Age: Part I-The Time Period Taken to Acquire First Words and First Word Combinations. Ear & Hearing. 2009, 30(5):526-540.
3. Nott P, Cowan R, Brown M, Wigglesworth G. Early Language Development in Children with Profound Hearing Loss Fitted with a Device at a Young Age: Part II-Content of the First Lexicon. Ear & Hearing. 2009, 30(5):541-551.
4. Schorr EA, Roth FP, Fox NA. A Comparison of the Speech and Language Skills of Children With Cochlear Implants and Children With Normal Hearing. Communication Disorders Quarterly. 2008, 29(4):195-210.
5. Ertmer DJ, Strong LM, Sadagopan N. Beginning to Communicate After Cochlear Implantation: Oral Language Development in a Young Child. Journal of Speech, Language & Hearing Research. 2003, 46(2):328.
6. Miyamoto RT, Hay-Mccutcheon MJ, Kirk KI, Houston DM, Bergeson-Dana T. Language skills of profoundly deaf children who received cochlear implants under 12 months of age: a preliminary study. Acta Oto-Laryngologica. 2008, 128(4):373-377.
7. Geers AE, Nicholas JG. Expected Test Scores for Preschoolers With a Cochlear Implant Who Use Spoken Language. American Journal of Speech-Language Pathology. 2008, 17:121-138.
8. Thal D, DesJardin JL, Eisenberg LS. Validity of theMacArthur-BatesCommunicative Development Inventories for Measuring Language Abilities in Children With Cochlear Implants. American Journal of Speech-Language Pathology. 2007, 16:54-64.
9. Rinaldi P, Caselli C. Lexical and Grammatical Abilities in Deaf Italian Preschoolers: The Role of Duration of Formal Language Experience. Journal of Deaf Studies and Deaf Education. 2009, 14(1):63-75.
10. Rescorla L, Achenbach T. Use of the Language Development Survey (LDS) in a National Probability Sample of Children 18 to 35 Months Old. Journal of Speech, Language & Hearing Research. 2002, 45(4):733.
11. Rescorla L, Alley A. Validation of the Language Development Survey (LDS): A Parent Report Tool for Identifying Language Delay in Toddlers. Journal of Speech, Language & Hearing Research. 2001, 44(2):434.
12. Rescorla L, Ratner N, Jusczyk P, Jusczyk A. Concurrent Validity of the Language Development Survey: Associations With the MacArthur-Bates Communicative Development Inventories: Words and Sentences. American Journal of Speech-Language Pathology. 2005, 14(2):156-163.
13. Capovilla FC, Capovilla AGS. Desenvolvimento Linguístico na Criança dos Dois aos Seis Anos: Tradução e Estandardização do Peabody Picture Vocabulary Test de Dunn & Dunn, e da Language Development Survey de Rescorla. Ciência Cognitiva: Teoria, Pesquisa e Aplicação. 1997, 1(1):353-380.
14. Kiese-Himmel C, Reeh M. Assessment of expressive vocabulary outcomes in hearing-impaired children with hearing aids: do bilaterally hearing-impaired children catch up? The Journal of Laryngology & Otology. 2006, 120:619-626.
15. Melo TM, Moret ALM, Bevilacqua MC. Avaliação da produção de fala em crianças deficientes auditivas usuárias de Implante Coclear Multicanal. Rev Soc Bras Fonoaudiol. 2008, 13(1):45-51.
16. DesJardin JL, Eisenberg LS. Maternal Contributions: Supporting Language Development in Young Children with Cochlear Implants. Ear & Hearing, 28(4):456-469.
17. ABEP - Associação Brasileira de Empresas de Pesquisa (2008). [homepage da Internet]. Acesso em 02 de julho de 2009. Disponível em: www.abep.org.
18. Bevilacqua MC, Yamada MO, Moret ALM. Critérios de Referência quanto ao Grau de Permeabilidade da Família no Processo Terapêutico. Centro de Pesquisas Audiológicas, Hospital de Reabilitação de Anomalias Craniofaciais, Universidade de São Paulo - CPA/ HRAC-USP, Agosto, 2001.
19. Takahashi RT, Fernandes MFP. Plano de aula: conceitos e metodologia. Acta Paul. Enf. 2004, 17(1):114-118.
1 Phonoaudiologist with Specialization in Choclear Implant from HC/FMUSP.
2 Doctoral Degree in Rehabilitation Department of Sciences Physiotherapy, Phonoaudiology and Occupational Therapy of the Medical School of the University of São Paulo.
3 Doctoral Degree in Otorhinolaryngology from FMUSP.
4 Titular Professor of the Medical School of the University of São Paulo.
5 Doctor Professor of the Medical School of the University of São Paulo.
6 Titular Professor of the Medical School of the University of São Paulo.
7 Associate Professor of the Medical School of the University of São Paulo.
Institution: Medical School of the University of São Paulo. São Paulo / SP - Brazil. Mail address: Associate Professor Débora Befi-Lopes - Rua Cipotânea, 51 - Cidade Universitária - São Paulo / SP - Brazil - Zip code: 05360-160 - Telephone: (+55 11) 3091-8419 - Email: dmblopes@usp.br
Article received on September 13, 2010. Approved on October 7, 2010.