INTRODUCTIONKeloids are benign fibrous tumors (1) that emerge as reactions to abnormal skin healing. Keloids develop due to the response of connective tissue to trauma and inflammation, burns, "piercing," or surgeries in areas of high tension in the body, in predisposed individuals (1, 2). Keloids may occur at any age regardless of gender; however, they tend to occur more frequently in individuals between 10 and 30 years of age. There is a higher prevalence in patients with dark skin and in the areas of the ear, shoulder, and chest (3).
The keloid appears as a thick and smooth (2) scar that extends beyond the original wound and may cause increased tension on the skin, leading to local deformation (3). Keloids usually start to grow within the first year after the trauma occurs, and they rarely regress on their own (2). The main symptoms associated with keloids are tingling, burning, and itching, leading to functional changes and discomfort (1).
At the cellular level, the characteristics of keloids include uncontrolled synthesis and deposition of excessive collagen and glycoproteins (2). It has been demonstrated that keloids are composed of polyclonal fibroblasts that are intrinsically normal, but exhibit abnormal responses to extracellular signals, thus resulting in excessive collagen production and decreased degradation of fibrotic scar tissue (1).
Differential diagnosis of keloids is made with hypertrophic scars, which are scars confined to the margins of the original wound and often show spontaneous regression (2). This distinction is very important, as these entities require different treatment strategies.
The retroauricular approach is widely used in otolaryngology, both in middle ear surgery (tympanoplasty, tympanomastoidectomy) or in the external ear (otoplasty) for either functional or purely aesthetic outcomes; this approach may result in a potential site of origin of keloids due to creating an area of high skin tension.
The objective of this study was to assess the epidemiology of patients with keloids (age, sex, skin color, procedure that led to the keloid) and the results of patients undergoing treatment of keloids after ear, nose, and throat (ENT) surgeries at our service center.
METHODThis study was approved by the Ethics Committee on Human Research of the HC/UFPR.
A chart review was performed for patients undergoing treatment of retroauricular keloids in the otorhinolaryngology service center, during the period of 2006 to 2009, and we prospectively evaluated patients undergoing keloid resection in 2010, with a protocol and evaluation of photos (pre- and postoperative).
We evaluated the type of surgery that resulted in the keloid, surgery for keloid resection, postoperative adjuvant treatments (beta-therapy or intralesional application of corticosteroids), number of recurrences and reoperations, characteristics of the recurrence (presence of redness, telangiectasia, coloring, and size of the scar), associated symptoms (pain and itching), and patient satisfaction about the outcome.
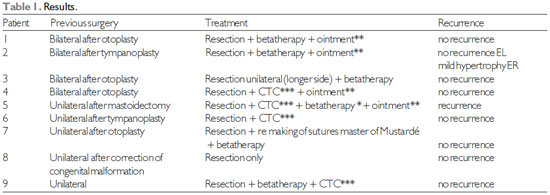
RESULTSWe evaluated 9 patients (13 ears), aged between 9 and 27 years, including 6 men and 3 women.
Four patients developed keloids after otoplasty, 2 after tympanoplasty, 1 after mastoidectomy, 1 after surgical correction of a congenital malformation (preauricular sinus), and 1 after a surgery that was performed at another institution (and thus cannot be described).
Patients underwent 6 sessions of beta-therapy at the Erasto Gaertner Hospital, and 2 patients had started taking corticosteroids and received beta-therapy late.
Two patients received only local corticosteroids (betamethasone 3mg, Celestone ® soluspan), and 1 patient did not undergo adjuvant therapy since the keloid was very small.
After the use of adjuvant therapies, we maintained the use of triamcinolone ointment 1 mg (Oncilom ®) with massage, for approximately 2 months.
The results are shown in Table 1 and Figures 1, 2, and 3.
The surgical technique is illustrated in Figure 4. The surgery was performed as follows: local anesthesia by infiltration of lidocaine 2% + aadrenaline 1:50,000, marking the margins of the keloid scar with gentian violet, creating a spindle-shaped incision, peeling the scar in layers, creating a sliding flap, hemostasis, suturing, and simple surgical wound dressing.
The beta-therapy was carried out with plates of Strontium (90Sr). The source of the beta-therapy was a 2-cm plate that was packed in a container, as shown in Figure 5. The instructions for use indicate that the board should stay out of the container only during the treatment period, the technician must keep as much distance as possible during the application, and the source of radiation has an acrylic plate that serves as a barrier to radiation. The patient is instructed to wash the area to be irradiated and dry the skin, and not to use any product or application during the irradiation period. The timing and amount of radiation in Grays varies with each patient, depending on the extent of the wound.
* Betatherapy started late (2 months)
** Corticosteroid triamcinolone ointment 1mg (Oncilom ®)
*** CTC = corticosteroid injection 3mg intralesional betamethasone (Celestone ® Soluspan)
Keloids are a problem that affects the quality of life of patients, not only for aesthetic reasons, but also primarily by the associated symptoms (itching, pain, burning sensation, and intolerance to some tissues) (4), making it necessary to utilize an effective, specific treatment approach.
The most common difficulty in treating keloids is their recurrence after surgical excision. To solve this problem, various types of adjuvant treatments have been developed.
The various treatment options used to prevent the recurrence of keloids include continuous pressure after surgery, application intralesional steroids, use of a carbon dioxide laser, application of silicone gel, administration of retinoic acid, covering with silicone gel (1), cryosurgery, use of chemotherapeutic agents (5-fluorouracil), intralesional interferon administration, radiation therapy, and others (5). However, there is no solid evidence that these methods are effective in prevention of keloid recurrence, and radiotherapy is the only treatment shown to be effective in preventing the formation of keloids (1).
It is important to remember that the keloid that is formed is not radiosensitive, but the fibroblasts in the recently scarred areas are; therefore, it is necessary to apply the radiation to the connective tissue in the immediate postoperative period, during the cellular proliferation and differentiation stage, and before the fibrotic tissue has formed (4).
The radiotherapy techniques that are typically employed include external radiotherapy with superficial X-rays, brachytherapy with strontium (Sr90) or electron beams, low-dose brachytherapy (1), high-dose brachytherapy (6,7), and radiation associated with pre or postoperative therapy (8).
The mechanism by which radiation acts is not well understood, but it is believed that radiation destroys fibroblasts, which are replaced by cells from the blood stream that migrate from other tissues (1).
Areas of excessive curvature of the skin can lead to greater difficulty in the delivery of postoperative radiation. Furthermore, the compression that is required for handling and radiating newly operated areas often is not well tolerated or may lead to damage to the areas that have been recently sutured. Huhne et al. (9) demonstrated a technique for postoperative radiotherapy that maintains the anatomical position of the ear, thus minimizing handling of the wound while maximizing the radiation dose to the target area. The external auditory canal is protected with a cotton ball and a lead shield, and the shell is filled with a moldable material (bolus Supertuff ®), thereby creating a flat and uniform level for the helix and tragus. Another material (Superflab®) is used to fill the retroauricular space, thus forming a continuous surface with the propeller and the antihelical fold. It is estimated that this arrangement would keep the target areas within 95% of the dose limit of the electron beam.
At our service center, we send patients who underwent keloid resection with radiotherapy to the Erastus Gaertner Hospital (oncology hospital) to undergo postoperative beta-therapy.
Beta-therapy is accomplished using plates of 90Sr (Strontium 90 radioisotope) in doses of approximately 30 Gy (Gray = unit of absorbed dose) (4).
According to HOCHMAN et al. (10), combining surgery with beta-therapy is the best treatment option, especially when the beta-therapy is administered 24 to 48 hours after the excision, in order to mitigate fibroplasia.
Our department obtained the best results from beta-therapy utilization when it was performed early (1 patient's beta-therapy was started the same day of surgery) in 4 patients. Only 1 patient received late beta-therapy (2 months after surgery), which resulted in relapse, even with early corticosteroid administration.
Postoperative percutaneous radiation therapy is associated with a few side effects, such as darkening of the skin in the treated area (11), but no serious adverse effects (such as the emergence of tumors) have been demonstrated, even in the long term (4,11).
Among the variety of technical aids that are available for the treatment of keloids, intralesional injection of corticosteroids (usually triamcinolone) is used for prevention or treatment of keloids in existing wounds. This administration may be intraoperative and postoperative, and has been shown to reduce the rate of recurrence of keloids (11); according to HOCHMAN et al. (10), it reduces keloid recurrence to less than 50%.
Bermueller et al. (12) achieved good cosmetic results and a high degree of patient satisfaction with resection, preparation of a skin flap, and retroauricular keloid complete closure with injection of corticosteroids to prevent formation of ear lobe keloids.
In our department, for the prevention of keloids after surgery, intralesional corticosteroids are used during the immediate postoperative period (1 mL per application, once every 2 weeks, for the first 2 months after surgery). We use 3-mg betamethasone (Celestone® soluspan). Although we still prefer beta-therapy, corticosteroid application is an alternative treatment when beta-therapy is not possible or not desired by the patient. In our study, 4 patients underwent application of corticosteroids after surgical resection, with good results.
The main side effects of corticosteroid therapy are local atrophy, depigmentation, and telangiectasia of the treated scar; although infrequent, some patients may be adverse to the use of systemic corticosteroids, even with the intralesional application. LIU and YEMCHA (13) reported a case of iatrogenic Cushing's syndrome that developed after the use of intralesional injections of triamcinolona for the treatment of multiple keloids. The authors suggest, therefore, that physicians consider the dose and number of injections of intralesional corticosteroids that are necessary for the treatment of keloids. Taking into account the potential for recurrence and the aggressiveness of disease, there may be a need for large volumes and frequency of injections, which increases the potential of systemic effects; thus, patients should have their adrenal function monitored before signs of the syndrome appear.
However, regardless of type of adjuvant used, the risk of recurrence depends on local factors that are related to surgical wounds and on general factors that are related to the patient. A higher recurrence of keloids is seen in areas in which the wound is sutured with tension, or there is postoperative infection and dehiscence of the suture. Patients with positive family history and previous history of recurrent keloids that have failed previous therapies have a higher risk of recurrence (10,11).
In our study, 1 patient did not undergo adjuvant therapy, but the keloid was small, showed no recurrence, and the patient was satisfied with the outcome.
The peak incidence of relapse occurs until around the 13th month, so patients with keloids should be monitored for a longer period of time (10). We routinely monitor our patients for at least 1 year after the completion of treatment.
Although keloids are more common in people of black ethnicity, all patients that were treated in our department were classified as Caucasians. According to HOCHMAN et al. (10), relapse rates show no relationship with ethnicity, age, or size of the lesion.
At the biochemical level, previous studies demonstrated increased expression of the cytokine transforming growth factor beta (TGF-) and its receptors in fibroblasts from keloid tissue. The TGF- family mediates physiological growth and promotes cell proliferation, inhibits apoptosis, induces synthesis of cyclooxygenase-2 (COX-2), and promotes tumor growth, invasion, and angiogenesis by upregulating the secretion of matrix metalloproteinase-2 (MMP-2). GAO et al. (14) showed increased expression of the gene for growth differentiation factor-9 (GDF-9, a family member of TGF-) in fibroblasts of the peripheral region of the keloid tissue, which is the area that is responsible for the growth and invasion of normal tissues. This result suggests that the fibroblasts that produce a GDF-9 are responsible for the invasion of peripheral tissue.
ULRICH et al. (15) showed that the formation of granulation tissue and wound healing depends on an adequate balance between the synthesis and degradation of the extracellular matrix, and that an excessive synthesis or degradation deficiency (or both) can lead to pathological scarring, including keloids. The enzymes involved in this process are the matrix metalloproteinases (MMPs), enzymes that degrade extracellular matrix components, which are inhibited by tissue inhibitors of metalloproteinases (TIMPs). The authors indicate that future non-surgical treatments that reduce the expression of TIMPs and increase that of MMPs may be effective therapeutic strategies for the treatment of pathologic scars such as keloids (15).
While these future therapeutic approaches may prove to be effective, the treatment of keloids, including retroauricular keloids, remains a challenge.
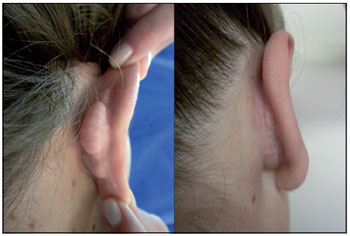
Figure 1. Case 3: Preoperative (left) and postoperative 3 months (right).
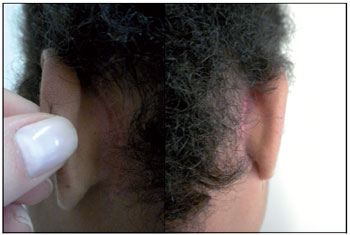
Figure 2. Case 2: Post-operative 3 months.
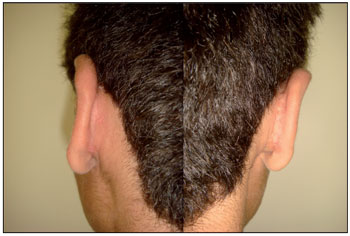
Figure 3. Case 1: Postoperative 1 year and 1 month.
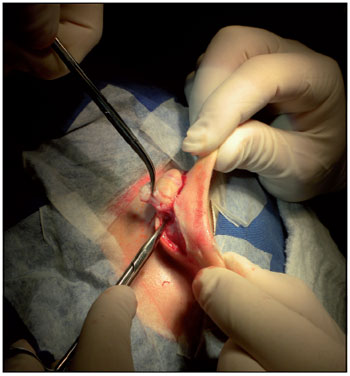
Figure 4. Surgical procedure - resection of the spindle keloid retroauricular flap by sliding.

Figure 5. Strontium container plate (betatherapy).
The treatment of retroauricular keloids presents many difficulties, either with regard to the treatment of keloid itself or due to location.
In our department, we obtained the best results when surgical excision was combined with beta-therapy that was started soon after surgery.
It is expected that the development of other non-surgical therapies will facilitate the treatment of retroauricular keloids and other keloids as well.
BIBLIOGRAPHIC REFERENCES1. Viani GA, Stefano EJ, Afonso SL, De Fendi LI . Posoperative strontium-90 brachytherapy in the prevention of keloids: results and prognostic factors. Int J Radiation Oncology Biol Phys. 2009, 73(5):1510-1516.
2. Froelich K, Staudenmaier R, Leinsasse N, Hagen R. Therapy of auricular keloids: review of different treatmente modalities and proposal for a therapeutic algorithm. Eur Arch Otorhinolaryngol. 2007, 264:1497-1508.
3. Correspondence and Comunication. Use of colchicine to prevent recurrence of ear keloids. A new approach. Journal of Plastic, Recontructive and Aesthetic Surgery. 2010, 63: e650-e652.
4. Oliveira Jr B, Lastoria JC, Pereira HR, Silveira LVA, Oliveira LP, Stolf HO. Estudo comparativo entre o tratamento radioterápico com elétrons e betaterapia, após cirurgia de quelóides. Surgical & Cosmetic Dermatology. 2009, 1(2):53-57.
5. Durani P, Bayat A. Levels of evidence for the treatment of keloid disease. Jouranl of Plastic, Reconstructive and Aesthetic Surgery. 2008, 61:4-17.
6. Garg MK, Weiss P, Sharma AK, Gorla GR, Jaggernauth W, Yaparpalvi R, Delrowe J, Beitler JJ. Adjuvant high dose brachytherapy (Ir-192) in the managemente of keloids which have recurred after surgical excision and external radiation. Radiotherapy and Oncology. 2004, 73:233-236.
7. Veen RE, Kal HB. Postoperative high-dose-rate brachytherapy in prevention of keloids. Int. J. Radiation Oncology Biol Phys. 2007, 69(4):1205-1208.
8. Stahl S, Barnea Y, Weiss J, Zaretski A, Leshem D, Miller E, Shafir R, Ben-yosef R, Gur E. Treatment of earlobe keloids by extralesional excision combined with preoperative and postoperative "sandwich" radiotherapy. Plastic and Reconstructive Surgery. January 2010,125(1).
9. Huhn JL, Johnson EL, St Clair W. Adjuvant radiation of bilateral postauricular keloids: an illustration of technique. Medical dosimetry 2007, 32(4):278-280.
10. Hochman B, Locali RF, Oliveira LQR, Ferreira LM. Disfunções cicatriciais hiperproliferaticas: Quelóide. Estima. 2004, 4(4):33-39.
11. Ragoowansi R, Cornes PGS, Glees JP, Powell BW, Moss LH. Ear-lobe keloids: treatment by a protocol of surgical excision and immediate postoperative adjuvant radiotherapy. British Journal of Plastic Surgery. 2001, 54: 504-508.
12. Bermueller C, Rettinger G, Keck T. Auricular keloids: treatment and results. Eur Arch Otorhinolaryngol. 2010, 267:575-580.
13. Liu MF, Yencha M. Cushing's syndrome secondary to intralesional steroid injections of multiple keloids. Otolaryngology-Head and Neck Surgery. 2006, 135:960-961.
14. Gao Z, Wu X, Song N, Zhang L, Liu W. Differential expression of growth differentiation factor-9 in keloids. Burns. 2010.
15. Ulrich D, Ulrich F, Unglaub F, Piatkowski A, Pallua N. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in patients with different types of scars and keloids. Journal of Plastic, Reconstructive & Aesthetic Surgery. 2010, 63:1015-1021.
1) Graduation in Medicine by UFPR /Federal University of Parana State, Brazil . Resident Doctor for Otolaryngology Service of HC/UFPR - Clinics Hospital/ Federal University of Parana State in Brazil.
2) ENT by ABORL/Brazilian Association of Otolaryngology and Cervicofacial Surgery. Otolaryngologist Doctor.
3) Academic of Medicine from UFPR/Federal University of Parana State, Brazil.
4) Master of Surgery by UFPR/Federal University of Parana State, Brazil. Responsible for the Skull Maxillofacial Surgery Sector of Otolaryngology Service of HC/UFPR - Clinics Hospital/ Federal University of Parana State in Brazil.
5) Ph.D. in Otolaryngology by Escola Paulista de Medicina (Medicine School of São Paulo State), Brazil (1986). Entitled Professor at Universidade Federal do Paraná / Federal University at Parana State, Doctor-in-Chief for Otolaryngology Service from HC/UFPR - Clinics Hospital/ Federal University of Parana State, Brazil.
Institution: Hospital de Clinicas da UFPR. Curitiba / PR - Brazil. Mailing address: Bettina Carvalho - General Carneiro Street, 181 - Centro, Curitiba / PR - Brazil - Zip-code: 80060-900 - E-mail: bettinacarvalho@yahoo.com.br
Article received in July 14, 2011. Article approved in September 19, 2011.