Bases Histopatológicas da Perda Auditiva em Recém-nascidos de Alto Risco
Histopathological Patterns of the Hearing Loss in
High Risk Neonates
Mônica Gondim Amatuzzi*, Clarinda Northrop**, Ricardo Ferreira Bento***, Roland D. Eavey****.
* PhD in Otorhinolaryngology by Faculdade de Medicina da Universidade de São Paulo (FMUSP).
** Head of Research of Temporal Bone Foundation, Boston.
*** Associated Professor of the Discipline of Otorhinolaryngology of Faculdade de Medicina da Universidade de São Paulo (FMUSP).
**** Professor of the Discipline of Pediatric Otorhinolaryngology of Massachusetts Eye and Ear Infirmary, Boston.
Study made at Massachusetts Eye and Ear Infirmary, Harvard Medical School, Boston as part of the doctorate thesis of the author.
Mail Address: Dra. Mônica Gondim Amatuzzi - Rua Dr. Eduardo de Souza Aranha, 99/82 - São Paulo / SP - CEP: 04543-120 - Phone: 55 (11) 3849-8859 - Fax: 55 (11) 3045-7301 - E-mail: mgamatuzzi@uol.com.br
This article was received on June 7, 2005, and accepted on June 13, 2005.
RESUMO
Introdução: A incidência de perda auditiva sensorioneural bilateral em recém-nascidos tratados em UTIs é de 2% a 4%. Vários fatores têm sido apontados na etiologia da perda auditiva, porém pouco se conhece a respeito das alterações histológicas da cóclea destes pacientes.
INTRODUCTIONThe incidence of sensorioneural hearing loss in newborns treated in Intensive Care Unit (ICU) is from 2% to 4%, what means 13 times larger than in normal newborns. (1,2).
The emerging of devices which facilitate newborn hearing screening (3) and the evidences that the early diagnosis of hearing loss is fundamental in language developing and social adaptation of these patients (4), have been calling the attention, in the literature, in identifying possible etiologic factors and in early diagnosing hearing loss.
The risk factors for hearing loss, such as congenital infection, neonatal anoxia, hyperbilirubinemia, use of ototoxics and presence of malformation of the head and neck have been intensely studied (5,6), causing conflicting data in relation to clinical condition and used treatments in ICU (7), therefore, knowledge in hearing loss physiopathology in these patients has been little developed.
The histopathologic studies, which could provide relevant data towards hearing loss physiopathology in newborns are not sufficient, due to difficulty in obtaining and in preparing temporal bones of newborns, specially the ones who were previously submitted to hearing evaluation.
The risk newborns are usually submitted to hearing evaluation little before hospital discharge. The patients in more severe conditions, who die, and who could have their temporal bones withdrawn for histological study, when submitted to autopsy, rarely have their hearing tested.
In this study, we described histopathologic findings of cochlea from 13 high-risk newborns who failed screening hearing exam made at ICU, and we also correlated such findings to clinical condition in patients.
MATERIAL AND METHODSMaterialIt was studied 26 temporal bones from 13 patients who were assisted and died in the ICU of Hospital Nacional de Niños (Child Hospital), São José, Costa Rica, from August 1991 to July 1992.
9 of those patients were males and 4 females, aging between 1 day and 2 months of life and weighing between 900 and 4000g. Seven of them were premature (less than 37 weeks of gestation age).
None of the mothers presented infection during pregnancy.
The most frequent clinical conditions among patients were: septicemia (77%), jaundice (54%), hyaline membrane disease (46%), anemia (38%), convulsion (31%), meconial aspiration syndrome (23%) and neonatal asphyxia (15%) (Graph 1).
Methods1. Electrophysiological Exam (BERA)The newborns were submitted to brainstem evoked response audiometry (BERA). Exams were done with a portable device, ALGO-1, which provides stimulus from 750 to 3.000 Hz, adjusted to intensity of 40 dB HL in order to eliminate possible conductive causes of hearing loss.
The device provides objective results as SUCCESSED, FAILED, COULD NOT BE TESTED, avoiding necessity of subjective interpretation of curves.
All patients studied failed the hearing exam.
2. Histopathologic Evaluation 26 temporal bones were withdrawn in the moment of autopsy, between 1 and 15 hours after death at Hospital Nacional de Niños, and were attached in formalin at 10%, decalcificated in tricholride acetic acid at 5% for about 40 days, dehydrated in increasing concentration of alcohol for 10 days and inserted in celloidin, according to the technique described by SCHUKNECHT (8). Blocks were cut in horizontal plan, of 20m thick, and in every 10 cuts one was colored with hematoxylin and eosin and used for the study.
The histological study was done at Massachusetts Eye and Ear Infirmary, Harvard Medical School, Boston (USA), and consisted of bidimensional reconstruction of cochleas by the technique of GUILD et al (9) and by evaluation of the following elements: number of ciliated cells and neurons, Reissner's membrane aspect, vascular wrinkle and spiral ligament and presence of blood in the interior of cochlea.
This study was approved by Comitê de Ética em Pesquisa (Ethics Committee) of Hospital Nacional de Niños, Costa Rica and by Massachusetts Eye and Ear Infirmary, Boston (Protocol: 0906-033).
RESULTSThe length of cochlear ducts determined by reconstructions ranged from 24 to 35 mm. The majority of them measured from 30 to 32 mm. The shortest duct was found in a patient with Trisomy 13 (the right duct measured 24 mm and the left one 25 mm).
1. Ciliated CellsIt was observed expressive loss of ciliated cells in 7 out of 13 patients studied. In all cases, this finding was bilateral and almost always symmetric.
Expressive loss of external ciliated cells was observed in two patients (Picture 1), in one of them, such loss was predominantly in basal spirals (Case 1) and in the other case at the top of cochlea (Case 2).
Selective loss of internal ciliated cells was observed in three patients (Picture 2). In two of the cases such loss was symmetric (Cases 4 and 5), and in the third case, loss was more extent in left cochlea than in the right one (Case 3).
The other two patients presented loss of internal and external ciliated cells predominantly in basal portion of cochlea, symmetric in Case 7 and more extent in right cochlea in Case 6.
From the other six patients, two presented normal histopathologic aspect, with excellent bilateral tissue preservation of cochleas (Cases 12 and 13) and in the other four patients, it was not observed any expressive loss of ciliated cells, though external ciliated cells presented round-shaped appearance oedema, and many times spotted from support cells, and in two of these cases (Cases 9 and 11), internal ciliated cells presented herniation of cell contents. Findings were symmetric in all cases.
Arrangements of histological alterations of ciliated cells are displayed in Graph 2.
2. NeuronsThe average number of neurons observed by cochlea was 21.959, which ranged from 13.596 to 29.566. The premature patients presented a number of neurons expressively inferior to non-premature patients (p< 0.05).
The patient having the least number of neurons was a premature baby with Trisomy 13, which presented shorter cochlea ducts (Case 6).
3. Reissner's MembraneOne of the patients with loss of internal ciliated cells (Case 3) presented Reissner's membrane jointed on Corti organ in bilateral way. None of the studied cases presented labyrinthic hydrops.
4. Stria vascularis and Spiral LigamentTwo patients presented deposit of basophilic material in stria vascularis in a bilateral way (Cases 2 and 9). None of them presented spiral ligament atrophy.
5. Intracochlear bloodIt was observed blood in perilymphatic space of cochlea in 6 patients, 5 was premature. In all cases, blood cells were placed in regions corresponding to the spiral ones, suggesting that their final localization is influenced by gravity powers.
All patients with blood in perilymphatic space had blood cells in the interior of cochlear aqueduct or next to its opening. The blood quantity ranged between the two sides and its presence did not show relation to Corti organ condition.
Table 1 presents a summary of clinical conditions and histopathologic findings from 13 patients studied.
DISCUSSIONHearing loss in the first years of life leads to a delay in language development and in social adaptation. Thus, Joint Committee on Infant Hearing (10) has established that hearing loss diagnosis should be done up to the first six months of life, so the child can be early inserted of prothesis and assisted by a specialized service.
Both BERA and otoemissions are considered efficient methods to evaluate newborn hearing, even if they are premature (11,12). According to some authors, BERA can also be a type of monitoring method of risk factors in newborns (13). In this study, the application of a portable device, which can be used in patients still in serious condition, enabled us to do, for the first time in the literature, the histological study of temporal bones of these people with early hearing evaluation.
The most frequent histological alteration in our study was the presence of blood in perilymphatic space, finding which confirms other literature reports in these people (14-17). Six patients presented different quantities of blood cells in perilymphatic space, possibly coming from cochear aqueduct. Therefore, localization of blood did not show special relation with other damaged areas of Corti organ, as it was also observed by HOLDEN AND SCHUKNECHT em1968 (18).
In relation to Corti organ, alterations in ciliated cells in 11 out of the 13 studied patients were observed. In the two patients with normal histological aspect, biochemical or morphologic alterations not detected at optic microscopy, or a disfunction in brainstem, could be responsible by test failure, as these tests were done in patients in serious condition.
The two patients with loss of external ciliated cells presented some clinical similarities: both were full-term babies, both had convulsions and took some of the medication in common though histological alteration reached different regions of cochleas in the two patients, predominantly on the base in one of the cases and apical in the other.
Three patients presented selective loss of internal ciliated cells, what is very uncommon. Usually, histological lesion of Corti organ starts from loss of external ciliated cells, developing with loss of internal ciliated cells, up to the comprehensive disappearance of Corti organ.
Selective loss of internal ciliated cells is described in animals, caused by some antineoplasic drugs (19,20), but in the literature there is only one case in humans, reported by SLACK AND col. (17), curiously, also in one premature newborn, as the three cases in our study.
In SLACK's study, authors related loss of internal ciliated cells to presence of a blood coagulum in the cochlea. Our results do not support this hypothesis, as one of the patients with loss of internal ciliated cells did not presented intracochlear blood, while the other premature patients with intracochlear blood did not presented loss of internal ciliated cells.
Hypothetically, selective loss of internal ciliated cells was already mentioned in the literature, by normal acoustic otoemission finding in the presence of hearing loss detected by audiometry of brainstem (21). Otoemmisions depend on normal functioning of external ciliated cells (22). In this way, a patient with loss of internal ciliated cells, but with no alteration of external ciliated cells, could, in a way, present normal acoustic otoemissions, even with an important hearing loss. This information is relevant, as the majority of services use otoemmisions as a way of screening in newborns.
Another important implication is that selective loss of internal ciliated cells cannot clinically differ from hearing neuropathy. In the two cases, the patient presented normal responses to otoemmisions and BERA alteration research. Apparently, the two cases are more common in premature babies. However, the prognosis of these patients in relation to cochlear implantation is very different, in other word, a patient with loss of ciliated cells, with no alteration of the hearing nerve would be a good candidate to cochlear implantation, on the contrary of the patient with hearing neuropathy. Perhaps this information justifies recent observations that some patients with clinical diagnosis of hearing neuropathy surprisingly develop well after cochlear implantation.
In the other six patients who presented loss of internal and external ciliated cells and morphological alteration of ciliated cells such as oedema, support cells displacement and herniation of cell content, histological alterations seem to occur in intensive way, without forming scars or tissue repairing. This type of histological finding is described in the process of "post-mortem" autolysis (23), what makes it difficult to trace correlations between histological findings and clinical data or screening hearing results, uncertain that these alterations occurred when patient was still alive.
CONCLUSION1. High risk newborns present different types of histological alterations in cochlea: loss of external ciliated cells, loss of internal ciliated cells, loss of internal and external ciliated cells and presence of intracochlear blood.
2. Premature condition is a relevant factor in cases of loss of internal ciliated cells.
3. Loss of internal ciliated cells can lead to errors in electrophysiological exam interpretation.
4. Differences of histological lesions suggest participation of more than on etiologic factor to hearing loss in high-risk newborns.
BIBLIOGRAPHY1. Stueve MP, O'Rourke C. Estimation of hearing loss in children: comparison of auditory steady-state response, auditory brainstem response, and behavioral test methods. Am J Audiol. 2003, 12(2):125-36.
2. Rhodes MC, Margolis RH, Hirsch JE, Napp AP. Hearing screening in the newborn intensive care nursery: comparison of methods. Otolaryngol Head Neck Surg. 1999, 120(6):799-808.
3. Clarke P, Iqbal M, Mitchell S. A comparison of transient-evoked otoacustic emissions and automated auditory brainstem responses for predischarge neonatal hearing screening. Int J Audiol. 2003, 42(8):443-7.
4. Pickard RE. Does early identification of deaf newborns lead to later improvements in language skills? JAMA 2002, 287 (5):587-8.
5. de Hoog M, van Zanten BA, Hop WC, Overbosch E, Weisglas_kuperus N, van den Anker JN. Newborn hearing screening: tobramycin and vancomycin are not risk factors for hearing loss. J Pediatr. 2003, 142(1):41-6.
6. Kountakis SE, Skoulas I, Phillips D, Chang CY. Risk factors for hearing loss in neonates: a prospective study. Am J Otolaryngol 2002, 23(3):133-7.
7. Eavey RD, Pinto LE, Thornton AR, Herrmann BS, Bertero MC, Saenz A. Early hearing testing of still critically-ill neonates. Arch. Otolaryngol. Head Neck Surg. 1996, 122:289-93.
8. Schuknecht HF. Pathology of the ear. 2nd edition. Philadelphia: Lea & Febiger, 1993, 1-29.
9. Guild SR. A graphic reconstruction method for the study of the organ of Corti. Anat. Rec. 22:141, 1921 apud Bredberg G., p.10, 1968.
10. Joint Committee on Infant Hearing; American Academy of Audiology; American Academy of Pediatrics; American Speech-Language-Hearing Association; Directors of Speech and Hearing Programs in State Health and Welfare Agencies. Pediatrics 2000, 106(4):798-817.
11. Dort JC, Tobolski C, Brown D. Screening strategies for neonatal hearing loss: which test is best? J Otolaryngol. 2000, 29(4):206-10.
12. Thornton AR, Kimm L, Kennedy CR. Methodological factors involved in neonatal screening using transient-evoked otoacustic emissions and automated auditory brainstem response testing. Hear Res 2003, 182(1-2):65-76.
13. Barden TP, Peltzman P. Newborn brain stem auditory evoked responses and perinatal clinical events. Am. J. Obstet. Gynecol. 1980, 136:12-9.
14. Buch NH. The inner ear of newborn infants (a histopathological study). J. Laryngol. Otol. 1966, 80:65-77.
15. Spector GJ, Pettit WJ, Davis G, Strauss M, Rauchbach E. Fetal respiratory distress causing CNS and inner ear hemorrhage. Laryngoscope 1978, 88:764-86.
16. Michaels L, Gould SJ, Wells M. The microslicing method in the study of temporal bone changes in the perinatal period: an interim report. Acta Otolaryngol. Suppl. (Stockh) 1985, 423:9-14.
17. Slack RWT, Wright A, Michaels L, Frohlich SA. Inner hair cell loss and intracochlear clot in the preterm infant. Clin. Otolaryngol. 1986, 11:443-6.
18. Holden HB, Schuknecht HF. Distribution pattern of blood in the inner ear following spontaneous subarachnoid haemorrhage. J. Otolaryngol. 1968, 82:21-9.
19. Takeno S, Harrison R.V, Ibrahim D, Wake M, Mount RJ. Cochlear function after selective inner hair cell degeneration induced by carboplatin. Hear. Res. 1994, 75:93-102.
20. Mount RJ, Takeno S, Wake M, Harrison RV. Carboplatin ototoxicity in the chinchilla: lesions of the vestibular sensory epithelium. Acta Otolaryngol. Suppl. (Stockh) 1995, 519: 60-5.
21. Bonfils P, Avan P, Londero A, Narcy P, Trotoux J. Progressive hereditary deafness with predominant inner hair cell loss. Am. J. Otol. 1991, 12:203-6.
22. Mountain D. Changes in endolymphatic potential and crossed olivocochlear bundle stimulation alter cochlear mechanics. Science 1980, 210:71-2.
23. Bagger-Sjöbäck D, Engström B. Preservation of the human cochlea. Ann. Otol. Rhinol. Laryngol. 1985, 4:84-92.
Graph 1. More frequent clinical conditions
Corti organ with loss of external ciliated cell (Case 1)
Corti organ with loss of internal ciliated cell (Case 3)
Histological aspect of Corti organ in 13 patients.
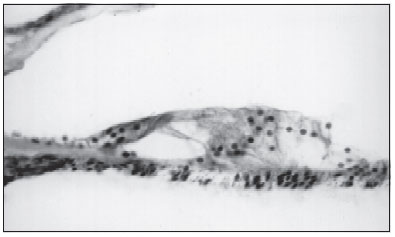
Picture 1
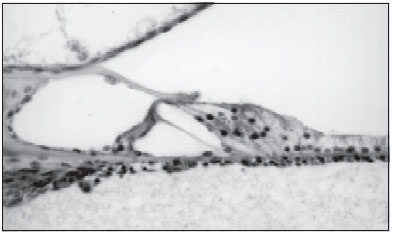
Picture 2